Polarity
Learn about electron affinity, electronegativity, and bond character in chemistry. Understand how polarity affects molecules and determine polar and nonpolar bonds. Practice analyzing different molecular structures to identify polarity.

Polarity
E N D
Presentation Transcript
Electron Affinity • A measure of the tendency of an atom to accept an electron • Increases with increasing atomic number within a period • Decreases with increasing atomic number within a group
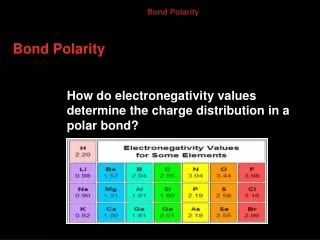
Electronegativity • Electronegativity – the relative ability of an atom to attract electrons in a chemical bond • Allows us to evaluate the electron affinity of specific atoms in a compound
Bond Character • A chemical bond between atoms of different elements is never completely ionic or covalent • Bond character depends on how strongly each of the bonded atoms attracts electrons
Polar Covalent Bonds • Results from unequal sharing of electrons in the bond • In a covalent bond, electrons spend more time near the nucleus of the atom with the higher electronegativity
Polarity • The atom that attracts electrons most strongly (has the highest electronegativity) has a slight negative charge • δ+ means partially positive • δ- means partially negative
Determining bond character • Look at HF • Electronegativity of F = 3.98 • Electronegativity of H = 2.20 • Difference: 3.98 - 2.20 = 1.78 • Electrons move towards the fluorine atom
Polarity • Polarity of molecules depends on • The presence of polar bonds • The position(s) of polar bonds in the molecule (polarity is a vector quantity) • Look at H2O and BeH2
Practice • Determine if the following molecules are polar • CH4 • CH2Cl2 • CHCl3 • CCl4 • CH3Cl
Nonpolar Polar Polar Nonpolar Polar