Oil Drop Experiment
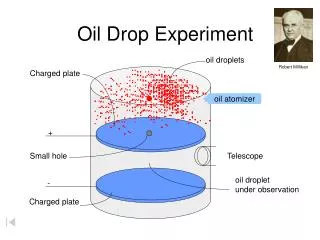
Oil Drop Experiment. How can we find the height of a molecule without fancy equipment?. Ben Franklin’s Oil Drop Experiment. Ben noticed that oil would spread out very thin….


Oil Drop Experiment
E N D
Presentation Transcript
How can we find the height of a molecule without fancy equipment?
Ben Franklin’s Oil Drop Experiment Ben noticed that oil would spread out very thin… “I fetched out a cruet of oil and dropped a little of it on the water. I saw it spread itself with surprising swiftness upon the surface… Though not more than a teaspoonful, produced an instant calm over a space several yards square which spread amazingly and extended itself gradually till it reached the lee side, making all that quarter of the pond, perhaps half an acre, as smooth as a looking glass.” Portrait of Benjamin Franklin by Joseph SiffredDuplessis Circa 1785
Let’s use these BBs to understand how we could find the height of a molecule. Imagine that each BB is one molecule. How can we find out how tall each one is, assuming we could not actually measure it? Image by HighPoint Learning
We know the volume of the BBs. Now, let’s pour them into a single layer. Image by HighPoint Learning
Measure the diameter of the single layer. If the shape were not a circle, how could you get a diameter? Given the diameter, calculate the Area. Area=π(Diameter/2)2 Image by HighPoint Learning
Now, let’s determine the height of this (1 layer thick) Cylinder. Use Height = Volume/Area.
Measure the height of a single BB to see How close our calculation is. ?
Now, you will use this same method to calculate the height of a single oil molecule. Image by Ben Mills
1) Prepare the oil and alcohol. • Mix 9 ml of alcohol with 1 ml of mineral oil. • Stir until they have combined well. • Each ml of this mixture will now be 1/10 ml oil.
2) Determine what fraction of a ml one drop is. • Fill the pipette to a ml mark and count how many drops it takes for the level to go down to the next ml mark. • How many drops are in a ml (e.g. 10 drops)? • What fraction of a ml is one drop(e.g. 1/10 ml)?
3) Calculate the volume of oil in the drop. • The volume of oil will be the 1/10 oil times the fraction of a ml that the oil drop (e.g. 1/10 x 1/10 = 1/100 ml of oil). • What is the volume of oil in one drop of the oil alcohol mix?
4) Prepare the pan of water. • Fill the pan with water. • Beat two chalkboard erasers together over the pan leaving a very thin film of dust on the surface.
5) Drop the oil drop. • Place the pipette of the oil alcohol mix close to the surface of the water in the middle of the pan. • Press one drop out onto the surface. It should push the chalk making a circle of oil that is easy to see.
6) Measure the diameter of the oil film. • Take several measurements of the diameter of the oil film and average them. • What is the average diameter of the oil film?
7) Calculate the area of the oil film • Area = π x (Diamerer/2)2 • What is the area of the oil film?
8) Calculate the height of the oil film • Height of the oil molecule = Volume of oil in the drop(from #3) / Area(from #7) • What is the height of the oil molecule? • What is the height of the oil molecule in nm?
This module is one of a series designed to introduce faculty and high school students to the basic concepts of nanotechnology. Each module includes a PowerPoint presentation, discussion questions, and hands-on activities, when applicable.The series was funded in part by:The National Science FoundationGrant DUE-0702976and the Oklahoma Nanotechnology Education InitiativeAny opinions, findings and conclusions or recommendations expressed in the material are those of the author and do not necessarily reflect the views of the National Science Foundation or the Oklahoma Nanotechnology Education Initiative.
Image Credits MilborneOne (Artists B787-2155a.jpg [Digital Image]. United Kingdom. Wikimedia Commons (commons.wikimedia.org) Mills, Ben. (Designer). Ball-and-stick model of the Oil Red O molecule. [Digital Image]. Wikimedia Commons. (commons.wikimedia.org) Molhave, Kristian (Professor. Fe-cat CVD MWCNT SEM overview mats with scalebar.jpg. [Scanning Electron Microscope Image]. United Kingdom. WIkimedi Commons (commons.wikimedia.org)
ReferencesEstimating the size of a molecule using an oil film (2004). Practical Physics. Retrieved from http://www.practicalphysics.org/go/Experiment_633.htmlFranklin, Benjamin. The Autobiography of Benjamin Franklin. [Kindle Edition]. Retrieved from http://www.amazon.com