Understanding Molecular and Structural Formulas with Resonance Structures
Explore the types of chemical formulas, including molecular, Lewis, and structural formulas. Learn how to determine center and terminal atoms, count valence electrons, and draw accurate bonding structures. Delve into resonance structures, where multiple Lewis structures can represent a molecule, and understand how certain molecules defy the octet rule. This guide covers examples like NH3, CO2, and PO4⁻³, with insights on molecules that exhibit odd valence electrons or expanded octets. Master the art of molecular drawing and deepen your knowledge of chemical bonding.

Understanding Molecular and Structural Formulas with Resonance Structures
E N D
Presentation Transcript
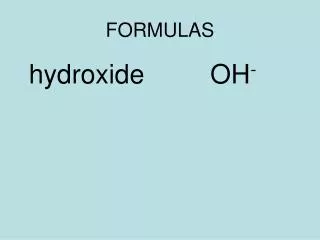
Types of formulas • Molecular formula = PH3 .. • Lewis structure = . P . .. • Structural formula = H - P – H I H
Hints for finding structural formulas • Decide if each atom is a terminal or center atom. • The true center atom is usually furthest left on the periodic table. • Add up the total number of valence electrons. • Fill in bonding pairs on trial structure • Count electrons left • Fill in lone pairs & check for electrons left • Adjust structure by adding double or triple bonds
Examples from 9.3 • NH3 • CO2 • PO4-3
Resonance Structures • Occurs when a molecule has a double & single bond • More than 1 Lewis structure possible • Only the bonds move, not atoms • Ex: NO3-1 • Molecule is really an average of all the possibilities • Bond length is like a 1.5 bond
Check for understanding • Draw Lewis structures for : CO3-2 S2O3 Which one has resonance? Which one does not show resonance?
Octet Rule Rebels • Molecules with an odd number of valence electrons • Molecules that form with fewer than 8 valence e- (B is a common rebel – often go on & form coordinate covalent bonds) • Molecules that form with more than 8 valence electrons (expanded octet) – uses d orbitals to do this, so period 3 or >. • All 3 types tend to be reactive & unstable.
Structures for rebels • Draw structures for: ClO2 NO BI3 BOBr SF6 XeF4