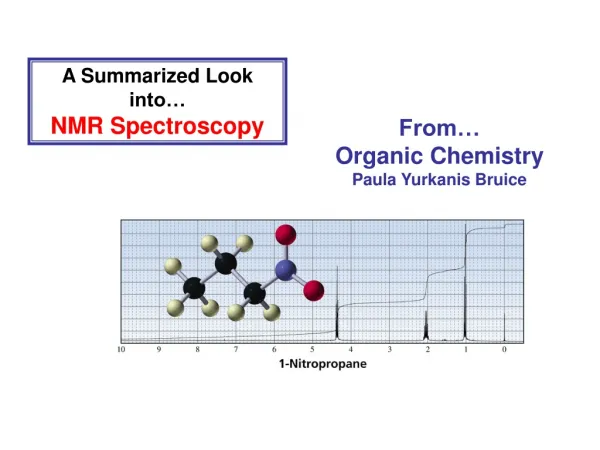
A Summarized Look into… NMR Spectroscopy
A Summarized Look into… NMR Spectroscopy. From… Organic Chemistry Paula Yurkanis Bruice. Nuclear Magnetic Resonance (NMR) Spectroscopy. Identify the carbon–hydrogen framework of an organic compound. Certain nuclei such as 1 H, 13 C, 19 F, and 31 P have

A Summarized Look into… NMR Spectroscopy
E N D
Presentation Transcript
A Summarized Look into… NMR Spectroscopy From… Organic Chemistry Paula Yurkanis Bruice
Nuclear Magnetic Resonance (NMR) Spectroscopy Identify the carbon–hydrogen framework of an organic compound Certain nuclei such as 1H, 13C, 19F, and 31P have allowed spin states of +1/2 and –1/2; this property allows them to be studied by NMR
The spin state of a nucleus is affected by an applied magnetic field
The energy difference between the two spin states depends on the strength of the magnetic field
absorb DE a-spin states b-spin states release DE Signals detected by NMR
The electrons surrounding a nucleus affect the effective magnetic field sensed by the nucleus
Chemically equivalent protons: protons in the same chemical environment Each set of chemically equivalent protons in a compound gives rise to a signal in an 1H NMR spectrum of that compound
The Chemical Shift The common scale for chemical shifts = d distance downfield from TMS (Hz) d = operating frequency of the spectrometer (MHz) The reference point of an NMR spectrum is defined by the position of TMS (zero ppm) The chemical shift is a measure of how far the signal is from the reference signal
1H NMR spectrum of 1-bromo-2,2-dimethylpropane
The chemical shift is independent of the operating frequency of the spectrometer
Electron withdrawal causes NMR signals to appear at higher frequency (at larger d values)
Characteristic Values of Chemical Shifts
1H NMR spectrum of 1-bromo-2,2-dimethylpropane
Integration Line The area under each signal is proportional to the number of protons that give rise to that signal The height of each integration step is proportional to the area under a specific signal The integration tells us the relative number of protons that give rise to each signal, not absolute number
Diamagnetic Anisotropy The p electrons are less tightly held by the nuclei than are s electrons; they are more free to move in response to a magnetic field Causes unusual chemical shifts for hydrogen bonded to carbons that form p bonds
Splitting of the Signals • An 1H NMR signal is split into N + 1 peaks, where N is the number of equivalent protons bonded to adjacent carbons • Coupled protons split each other’s signal • The number of peaks in a signal is called the multiplicity of the signal • The splitting of signals, caused by spin–spin coupling, occurs when different kinds of protons are close to one another
1H NMR Spectrum of 1,1-Dichloroethane
The ways in which the magnetic fields of three protons can be aligned
Splitting is observed if the protons are separated by more than three s bonds Long-range coupling occurs when the protons are separated by more than three bonds and one of the bonds is a double or a triple bond
The three vinylic protons are at relatively high frequency because of diamagnetic anisotropy
The Difference between a Quartet and a Doublet of Doublets