Covalent Bond
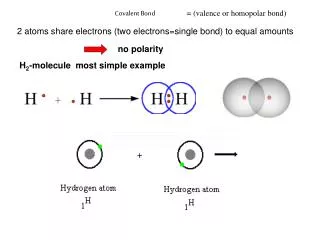
Covalent Bond. Covalent bonding. When atoms tend to attain octet arrangement by sharing , a bond will be formed between the sharing atoms . This type of bonding is called covalent bonding. 1. Sharing of one pair of electrons-Single bond 2. Sharing of two pairs of electrons-Double bond


Covalent Bond
E N D
Presentation Transcript
Covalent bonding When atoms tend to attain octet arrangement by sharing, a bond will be formed between the sharing atoms. This type of bonding is called covalent bonding.
1. Sharing of one pair of electrons-Single bond 2. Sharing of two pairs of electrons-Double bond 3. Sharing of three pairs of electrons-triple bond A covalent bond may be formed by . Single Bond or H-H
Oxygen exist as a molecule of two oxygen atoms. These two oxygen atoms share two pairs of valence electrons (four valence electrons total) between them, forming a DOUBLE COVALENT BOND. This is true of any double covalent bond; four valence electrons are shared
Nitrogen exists as a molecule made up of two nitrogen atoms that share three pairs of valence electrons (six valence electrons total) to form a TRIPLE COVALENT BOND.
The following animation shows the sharing of electrons (represented by Bone) equally between chlorine atoms. (represented by dog)
Properties of covalent compounds 1. Compounds containing covalent bond are made of molecules (not ions). 2. Low melting and boiling points 3. Poor electrical conductors in all phases 4. Many soluble in nonpolar solvents but not in water.