The Covalent Bond and Naming
280 likes | 563 Vues
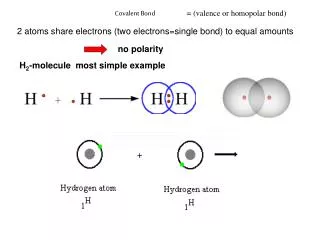
The Covalent Bond and Naming. Chapter 20. What is a Covalent Bond?. Covalent bond - bond that results from the sharing of electrons. - occurs between nonmetallic elements. Molecule - compound formed when two or more elements bond covalently. Formation of a Covalent Bond. H. H. H. H.

The Covalent Bond and Naming
E N D
Presentation Transcript
The Covalent Bond and Naming Chapter 20
What is a Covalent Bond? Covalent bond - bond that results from the sharing of electrons - occurs between nonmetallic elements Molecule - compound formed when two or more elements bond covalently.
Formation of a Covalent Bond H H H H Single covalent bond - two electrons shared H H - shown as a dash
Draw the electron dot structure for water, H2O O H O H H H
Double bond - sharing of two pairs of electrons between two atoms http://dkreutz.basd.k12.wi.us/A17-LewisDot-Octet.html Triple bond - sharing of three pairs of electrons between two atoms
Naming Binary Molecular Compounds Second element named with prefixes and -ide ending First element named with prefixes (except mono) Prefixes tell number of atoms of each element
Prefixes Used mono - 1 octa - 8 di - 2 nona - 9 tri - 3 deca - 10 tetra - 4 penta - 5 hexa - 6 hepta - 7
Name the compound P2O5 diphosphorus pentoxide • Your Turn!! • CCl4 • PBr3 • N2S3 carbon tetrachloride phosphorus tribromide dinitrogen trisulfide
Writing Formulas from Names Binary Molecular Compounds - use prefixes to give subscripts for each element What is the formula for dichlorine monoxide? Cl2O
Practice • Dihydrogen monoxide • Sulfer hexaflouride • Biboron tetrahydide • Carbon dioxide H2O SF6 B2H4 CO2
Ionic compounds vs. Covalent Molecules The differences and similarities
Ionic Compounds vs. Covalent Compounds • Ionic Compounds • Made up of positive metallic ions and negative nonmetallic ions after metal transferred electrons to nonmetal • Arranged in regular repeating patterns called crystals
Form crystals where every (+) ion is surrounded by (-) ions and every (-) ion is surrounded by (+) ions • Known as crystalline compound
Have high melting and boiling points • Conduct electricity when dissolved in water---electrolyte
Covalent Compounds • Made up of nonmetals sharing electrons • Liquids, gases, or solids • Have low melting and boiling points • Do not conduct electricity
Binary Ionic Compounds: Type I • Binary (Metal + Non-metal) Ionic Compound • Metal has a fixed charge • Example: • Ag+ • Zn2+ • Cd2+ • Al3+ • All alkali and alkaline earth metals
Steps to naming Binary Ionic compounds (Type I) Example: CaS • Write the name of the first element • Write the name of the second element with an –ide ending calcium sulfide calcium sulfide
Practice aluminum chloride calcium oxide lithium nitride magnesium iodide silver sulfide • AlCl3 • CaO • Li3N • MgI2 • Ag2S
Type I: Name to Formula Rb +1 P -3 Rb3P Write the ion of the metal and the non-metal Criss-cross the charges Example: rubidium phosphide
Naming Type IV Compounds Not a binary compound; therefore, it contains a polyatomic ion Polyatomic ions are groups of elements covalently bound (share electrons) that have a charge.
Polyatomic Ions Ammonium - NH4+1 nitrate - NO3-1 hydroxide - OH-1 phosphate - PO4-3 chlorate - ClO3-1 acetate - C2H3O2-1 carbonate - CO3-2 sulfate - SO4-2
Lets Try It!! Writing Names from Formulas 1.) NH4NO3 ammonium nitrate 2.) (NH4)3PO4 ammonium phosphate 3.) Mg(OH)2 magnesium hydroxide
Example • NH4NO3 • K2SO4 ammonium nitrate potassium sulfate
Type IV Practice sodium nitrate calcium sulfate ammonium oxide magnesium phosphate lithium acetate ammonium nitride • NaNO3 • CaSO4 • (NH4)2O • Mg3(PO4)2 • LiC2H3O2 • (NH4)3N
Name to formula: Type IV • Write the ions of each • Criss-cross the charges Ex: potassium sulfate? ammonium nitride? K+1 SO4-2 NH4+1 N-3 K2SO4 (NH4)3N