The Atom and Radiation
The Atom and Radiation. Nuclear Radiation. Goals: To answer the following questions. What is radiation? Are there different types? Are all forms (equally) dangerous? Where is radiation found? Is all radiation man-made? What is radiation used for? Are there benefits to some types?

The Atom and Radiation
E N D
Presentation Transcript
The Atom and Radiation Nuclear Radiation
Goals: To answer the following questions • What is radiation? • Are there different types? • Are all forms (equally) dangerous? • Where is radiation found? • Is all radiation man-made? • What is radiation used for? • Are there benefits to some types? • How long is something radioactive for?
The Discovery of Radioactivity • The German physicist W.K. Roentgen “accidentally” discovers a mysterious source of radiant energy that can pass through low density shields (like card board). He calls this mysterious energy X-rays. • Further research showed that X-rays cannot pass through everything, particularly high density materials like lead and bone. • Roentgen takes the first X-rays of his wife’s hand to present to his colleagues.
The Discovery of Radioactivity • The French physicist Henri Becquerel takes interest in Roentgen's X-rays in 1896. He investigated whether certain minerals could emit X-rays. He experiments with Uranium and a photographic plate (develops upon exposure to light). • Another accident happens…Becquerel becomes frustrated with his research, wraps the photographic plate in black paper (to prevent light exposure), throws it in his desk drawer with a piece of Uranium on top and closes it up. • What do you know??? A few days later, Becquerel discovers that the photographic plate has been exposed while sitting in his dark drawer.
The Discovery of Radioactivity • What Becquerel inadvertently discovered was radioactivity, the spontaneous emission of nuclear radiation. • Soon after (in 1898,) Becquerel's colleagues, Marie Curie and her husband Pierre discover two other radioactive elements: polonium and radium.
What is Radioactivity? • Radioactivity: the spontaneous emission of nuclear radiation. • We now know that there are two categories of radiation: • Non-ionizing radiation – low-energy radiation that transfers energy to matter • usually only harmful in large amounts • Ionizing radiation – high-energy radiation that can eject electrons from atoms/molecules to form highly reactive ions and can cause serious cell damage • exposure should be limited.
Concerns….. • But radiation is all around us…the question is, should we be concerned about our safety?? Are we in danger of serious exposure to radiation? Can we use radiation?
Forms of Radiation • Radiation comes in several forms as shown in the electromagnetic spectrum below; but not all forms are represented here
Types of Radiation • Three main types (from the 2 categories) : 1. Non-ionizing electromagnetic radiation • Radio • Micro • Infrared • Visible • low energy UV
2. Ionizing electromagnetic radiation • High UV • X-rays • Gamma rays 3. Ionizing atomic particle radiation • radioactive elements
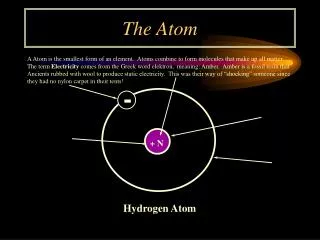
Why are some elements radioactive? To answer this question, you must understand a little about atomic structure. • All matter can be broken down into atoms:
An atom is composed of three parts: • Protons • Neutrons • Electrons
Isotopes (cont.) • Some isotopes are stable and others are unstable. This is where radioactivity comes in. • A stable isotope is not radioactive, but an unstable isotope is! • Ex. 12C is stable • 13C is stable • 14C is radioactive • Radioactive elements will emit radiation until they become a stable isotope.
Every Element has Isotopes – the amount of each isotope is fixed • Ex. Uranium • Which isotope of Uranium is used to make an atomic bomb?
Emitted radiation • This emitted radiation can be one of three types: • Alpha ( 4α or 4 He ) • heavy particle radiation • easily blocked because its so big, but • the most dangerous particle • Beta ( 0β or 0 e) • particle radiation smaller than alpha 2 2 -1 -1
Emitted radiation • This emitted radiation can be one of three types: • Positron ( 0β or 0 e ) • positive beta radiation • Gamma ( 0 γ ) • high energy radiation +1 +1 0
Emitted radiation • Alpha and Beta emission cause radioactive elements to change to a new element. • Gamma causes no change in the radioactive element.
Radiation Exposure • Naturally occurring radioisotopes provide a constant small dose of radiation • Radioactive isotopes constantly decay, releasing alpha, beta and/or gamma radiation. • This constant, inescapable radiation is called background radiation.
Natural background radiation: - Outer space • All forms of electromagnetic radiation - Ground water, rocks, soil • contain Uranium and Thorium - Atmosphere • contains radon - Food and Environment • like C-14 and potassium
Radon • Produced as Uranium in the soil decays. Uranium decays to produce radon gas: • When this gas is inhaled, it further decays in your lungs into Polonium, Bismuth and Lead (these heavy metals cannot be exhaled). • The resulting alpha radiation is being released into your body, causing cell damage.
Manmade background radiation • Fallout (nuclear weapons testing) • Airplane flights • Released from • burning fossil fuels • nuclear power plants • mining • making • cement • concrete • sheet rock
Medical Applications • Medical uses of radioisotopes fall into two categories. • Diagnostic • Therapeutic
Diagnostic • A standard x-ray cannot produce an image of an organ like the heart, liver, pancreas, blood vessels, etc. • To illuminate the targeted region, a radioisotope is injected into the body. • Radioisotopes used are low dosages with a short half-life. • localized 0.1 to 50 rem doses are common
Commonly Used Radioisotopes • Americium-241= Diagnose thyroid disorders, smoke detectors. • Cesium-137= Cancer treatment. • Iodine-125,131= Diagnosis & treatment liver, kidney, heart, lung and brain. • Technetium-99=Bone and brain imaging; thyroid and liver studies; localization of brain tumors.
CAT & PET scans • A computerized tomography scan is an enhanced x-ray machine using multiple beams capable of producing a 3D image. • A positron emission tomography scan uses a positron emitter to generate a 3D image.
MRI • In magnetic resonance imaging, a powerful magnetic field is used along with low energy radio frequency to generate an image. • Based on the premise that hydrogen atoms have “spin.” • In a powerful magnetic field, these hydrogen atoms can be made to flip between the two spin states. • THIS IS NOT NUCLEAR RADIATION
Therapeutic • In radiation therapy, radiation is used to target cancer cells. • Radiation levels are in very high dosages. • localized 4000 – 6000rem doses are common • Cyberknife treatment uses computer technology to aim 100’s of x-ray beams precisely at a tumor.
Measuring Radiation • Mainly, two units are used to measure radiation: • RAD = radiation absorbed dosage • 1 RAD = 1.0 x 10-2 J / kg of body tissue • RBE = a multiplier for each type of radiation. • alpha = 20; protons, neutrons = 10; betas, positrons, and gammas = 1. • REM = radiation equivalent in humans • REM = RAD x RBE • There are several other units out there; we are not going to worry about those.
Biological Effects • Alpha particles cannot penetrate our skin, yet internally they can cause massive damage on soft tissues like the lungs and intestinal linings. • Beta particles can cause a burn on the outer portion of the skin. • Gamma particles penetrate completely and if exposed to large quantities is deadly.
Biological Effects • Radiation affects those cells in our body that undergo rapid cell division like bone marrow, intestinal lining, and the skin. • Radiation tends not to affect cells that remain unchanged like our brain, liver, muscles, etc. • Doesn’t mean it CAN’T, it just is less likely
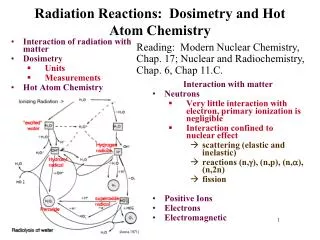
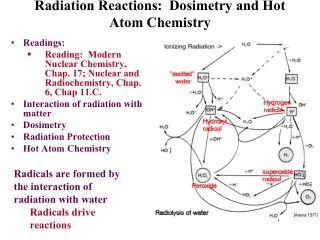
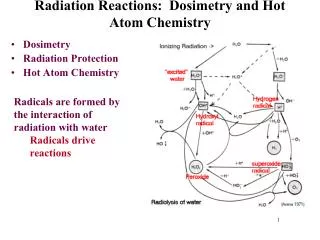
How It Works… • When radiation hits a molecule like water, it ionizes. • This water molecule reacts with another water molecule.
How It Works… • The “dot” on the OH is an odd electron. • Molecules with an odd electron are called free radicals. • Free radicals are electron scavengers and interfere with electron transfer reactions – many of which are vital in the function of the body.
How much is safe? • Ionizing radiation breaks bonds in molecules within the body. At low exposure levels, your body can fix the minimal damage. Higher exposure levels that your body cannot fix will lead to damaged DNA, causing mutations (tumors and birth defects)
Average U.S. individual receives 0.360 rem per year. • About 0.300 rem of this is from natural sources • U.S. limit for background radiation in a given area is 0.500 rem. • U.S. safe exposure in the work environment is 5.000 rem.
Dose Response Relationships • 0-15 rem—No or minimal symptoms • 15-40 rem—Moderate to severe illness • 40-80 rem—Severe illness deaths start above 50 rem • Above 80 rem—Fatal ***Acute whole body doses
Activity Typical Dose Smoking 280 millirem/year Radioactive materials use in a UM lab <10 millirem/year Dental x-ray 10 millirem per x-ray Chest x-ray 8 millirem per x-ray Drinking water 5 millirem/year Cross country round trip by air 5 millirem per trip Coal Burning power plant 0.165 millirem/year Your Annual Exposure
Radiation Protection • Decrease Time • Increase Distance • Increase Shielding
Measurement and Detection • A Geiger tube is often used to detect radiation. • Consists of a metal tube filled with a gas like Argon. • When radiation enters the tube, it ionizes the gas. • The ions are attracted to a negative charged wire and the electrons are then counted.
Measurement and Detection Animation: http://www.dlt.ncssm.edu/tiger/Flash/nuclear/GeigerTube.html
Alpha • Radioactive isotopes decay until a stable nucleus can be formed. What happens when radioactive isotopes decay? • Many elements release alpha radiation: 226 Ra 4 He + 222 Rn (radium)(alpha particle) (Radon) • As these radioactive isotopes decay, an alpha particle is released and a new element is formed. 2 86 88
Alpha Particles • Alpha Particles: 2 neutrons and 2 protons • They travel short distances, have large mass • Only a hazard when inhaled or formed in the lungs • because they can’t pass through skin)
Beta Many elements release beta radiation: 222 Rn 0 e + 222 Fr (radon)(neg. beta particle) (Francium) • As these radioactive isotopes decay, a beta particle is released and a new element is formed. -1 86 87