Bonding
Bonding. Chemistry revision. There are two types of bonding you need to be familiar with…. Covalent bonding Ionic bonding. You will be re-visiting…. The structure of the atom. Why atoms want to lose or gain electrons. Which type of materials use covalent or ionic bonding.


Bonding
E N D
Presentation Transcript
Bonding Chemistry revision
There are two types of bonding you need to be familiar with… • Covalent bonding • Ionic bonding
You will be re-visiting… The structure of the atom. Why atoms want to lose or gain electrons. Which type of materials use covalent or ionic bonding. The properties that covalent and ionic bonding give to the material.
You need to be able to… Name the components of an atom. Understand how many electrons are in each orbital. Describe the charge on each sub atomic particle. Understand what an isotope is. To draw dot and cross diagrams. To draw ionic formula diagrams. To name compounds which use these types of bonding
The atom Atoms contain threesub-atomic particles called protons, neutronsand electrons. The protons and neutrons are found in the nucleus at the centre of the atom, and the electrons are arranged in shells around the nucleus.
The charge in an atom • Atomic heaven is to be neutral! • Atoms will share or steal electrons so they can to obtain the structure of the Group 0 or Noble elements. • This group reacts with very little at all! • By sharing or stealing electrons the atoms bond to become compounds and therefore neutral!
There are two properties of sub-atomic particles that are especially important:MassElectrical charge
So now we look at the Periodic Table… Here is the elementCarbon Atomic Number = the number of protons in the nucleus.It is the smaller number in the box. Atomic Mass = the number of protons and neutrons in the nucleus.It is the larger number in the box. 12 C 6 From these two numbers we can work out the number of electrons this element should have.
Carbon can also be an isotope Isotopes have the same number of protons but a different number of neutrons. So isotopes have the sameatomic number but a different atomic mass. 126 C or 146 C Other famous isotopes are uranium and radium
Where are the electrons? The electrons are located in orbitals or shells around the nucleus. The innermost orbital will take up to two electrons only. The next three orbitals will take a maximum of eight electrons each.
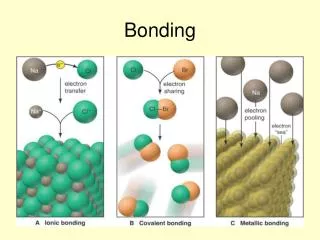
Covalent Bonding • Covalent compounds are formed when non-metal atoms react together. • As these atoms come near their outer electrons are attracted to the nucleus of both atoms and become shared by the atoms. • The shared electrons count towards the shells of both atoms and therefore help fill up incomplete electron shells. Boardworks – Covalent Bonding
Examples of covalent bonding Simple covalent bonds: Water H2O Ammonia NH3 Methane CH4 Oxygen O2 Giant covalent structures: Graphite Diamond
Properties of covalent compounds Small numbers of atoms in the molecules. Low melting and boiling points. Most are gases or liquids at room temperature. They do not conduct electricity. They tend to be insoluble in water.
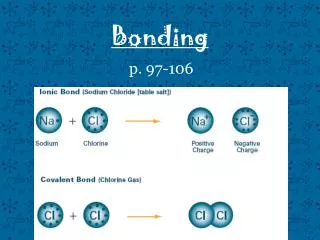
Ionic bonding Most ionic compounds contain a metal and a non-metal. When metals react they lose outer shell electrons to leave a full electron shell. This produces a charged atom (ion) with a + charge. When non-metals react with a metal they gain electrons to achieve a full electron shell.
Examples of ionic structures http://www.chem.ox.ac.uk/icl/heyes/structure_of_solids/Movies/NaCl.html Animation of NaCL
Properties of ionic compounds Atoms are held very strongly together. The compound has a regular structure. Ions form giant structures. Very high melting and boiling points. Soluble in water. Conducts electricity when dissolved.
OK, have a go at this quiz and check what you should know http://www.sciencepages.co.uk/keystage4/GCSEChemistry/module3.php
This is the structure of Argon It’s electronic configuration is written 2,8,8 Which group of the Periodic Table will it be found in?