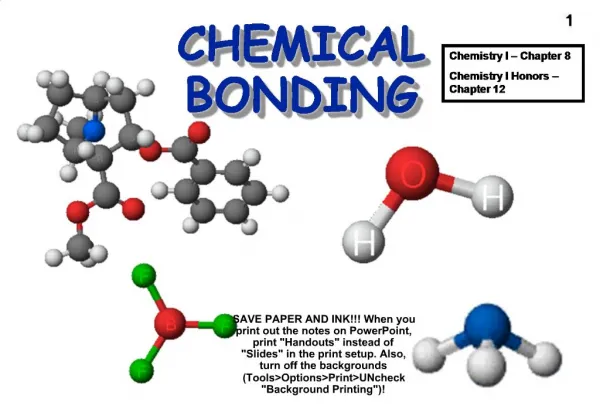
Chemical Bonding
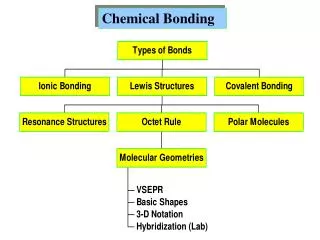
Chemical Bonding. Ionic and Covalent. Chemical Bond. Forms between atoms if their valence electrons make a new arrangement that has less energy then their previous arrangement (more stable together than separate) Atoms can lose, gain or share electrons to form bonds. Review: Octet Rule:.

Chemical Bonding
E N D
Presentation Transcript
Chemical Bonding Ionic and Covalent
Chemical Bond • Forms between atoms if their valence electrons make a new arrangement that has less energy then their previous arrangement (more stable together than separate) • Atoms can lose, gain or share electrons to form bonds
Review: Octet Rule: • When atoms combine, bonds form so that each atom finishes with an octet (8) valence electrons
Review: Ionic Bonds: • Between cations (+) and anions (-) • Is an attraction between oppositely charged ions • Metal ion “gives” electrons to non-metal ion • Ex : NaCl Na “gives” one e- to Cl
Covalent Bonds • Between 2 non-metal ions • Share one or more pairs of electrons • **A neutral particle that is composed of atoms joined by covalent bonds is called a Molecule. • Substances that are composed of molecules are called Molecular Compounds.
Covalent bonding • A single bond is formed when one pair of electrons is shared. • In a double bond 2 pairs and in a triple bond 3 pairs of electrons are shared • Example: diatomic molecules, like O2, H2, N2, F2, Cl2…
Diatomic molecules • A diatomic molecule is a molecule made of two of the same atoms. • Hydrogen exists as a diatomic molecule. • It bonds with itself to form the molecule H2. Electron dot diagram: H· + ·H H:H
Other diatomic molecules: • A total of seven elements exist as diatomic molecules rather than as individual atoms. • They are; H2, N2, O2, F2, Cl2, Br2, I2 • Remember; “HOBrFINCl”….hehe
Lewis structures • also called Lewis-dot diagrams • are diagrams that show the bonding between atoms of a molecule and other electrons that may exist in the molecule.
Representing Covalent Molecules: • No Arrows! • Use circles to show shared pairs of electrons • Draw electron dot diagrams side by side and see if pairs of electrons can be shared. • Remember one electron in pair must come from each atom.
Br2 O2 N2 CH4 SiH4 CBr4 CBr2F2 H2O CO2 H2S GeH4 C2H2 Examples: