Classifying matter
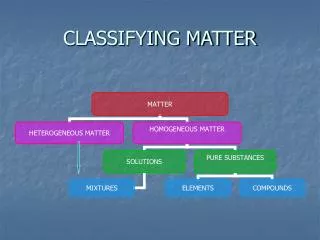
Classifying matter. Ch. 2. Pure Substances. - matter that always has the same composition Every sample of the substances always has to same properties Uniform composition, fixed properties. Elements. Cannot be broken down into simple substances

Classifying matter
E N D
Presentation Transcript
Classifying matter Ch. 2
Pure Substances • - matter that always has the same composition • Every sample of the substances always has to same properties • Uniform composition, fixed properties
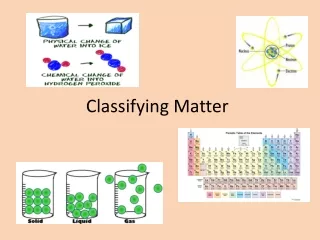
Elements • Cannot be broken down into simple substances • Contains only 1 type of atom(smallest particle of an element) • Symbols • 1st letter always capitalized, 2nd never • Same globally • Ex:
Compounds • Substance made of 2 or more simpler substances • Can be broken down into simpler substances • Properties differ from the elements in which its made • Has a fixed proportion • Ex:
Mixtures • Retain some properties of individual substances • Composition is not fixed
Mixtures Heterogeneous Homogenous • Different types of substances • Unevenly mixed • Appears to be only 1 substance • Evenly mixed
Solution • Homogenous Mixtures • Particles are too small- to settle out, to be trapped by filter, or scatter light • Ex:
Suspension • Heterogeneous Mixtures • Separates into layers over time • Particles are trapped by filters, and can scatter light • Ex:
Colloid • Heterogeneous Mixture • Particles never settle out • Scatter light= Tyndall Effect • Ex:
Physical properties Ch. 2.2
Physical Properties • Characteristics of matter • Observed/ measured without changing the composition
Physical Properties Viscosity Conductivity • Resistance to flow • High viscosity= slow • Low viscosity= fast • Decreases with heat • Ability for heat to flow • High conductivity= conductors
Physical Properties Malleability Hardness • Ability to be hammered without shattering • Ability to scratch another surface • Diamond- hardest known material
Physical Properties Melting/Boiling Points Density • Temp. which solid turn liquid- melting point • Temp. which liquid turns gas- boiling point • Test purity of a substance
Using Physical Properties • ID Materials • 1-decide which properties to test • 2-test the sample of unknown • 3- compare results to known • Choose Materials • Look at properties for specific uses
Using Physical Properties • Filtration • Process that separates materials based on particle • Distillation • Process that separates substances based on boiling points
Physical Change • Properties change • Substances materials remain the same!
2.3 Chemical Properties
Chemical Properties • Ability to produce a change in the composition of matter • 1 substance changes into another
Chemical Properties Flammability Reactivity • Ability to burn in presence of oxygen • How readily a substance combines chemically with other substances
Chemical Change • 1 substances reacts with another to form 1 or more new substances
Signs of Chemical Change • Color Change- • Production of Gas- • Formation of Precipitate-