Respiration
490 likes | 659 Vues
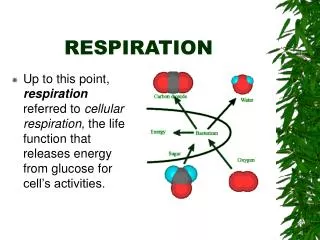
Respiration. Chapter 9. Energy and Living Things. ENERGY FOR CELLS. Where do cells get their energy? How do cells get energy?. TWO TYPES OF CHEMICAL REACTIONS. Endergonic – energy is absorbed. Energy of the products is greater than the energy of the reactants. Uphill.

Respiration
E N D
Presentation Transcript
Respiration Chapter 9
ENERGY FOR CELLS Where do cells get their energy? How do cells get energy?
TWO TYPES OF CHEMICAL REACTIONS • Endergonic – energy is absorbed. Energy of the products is greater than the energy of the reactants. Uphill
Exergonic – Energy is released. Energy of the products is less than the energy of the reactants. Downhill
Endergonic Reaction • X-axis=time • Y-axis=energy • Still requires activation energy P R
Exergonic Reaction • X-axis=time • Y-axis=energy • Still requires activation energy R P
ENERGY FOR CELLS • All living things get energy from the sun either directly or indirectly.
Plants use the sun’s energy directly to make food • Sunlight energy must be changed to energy trapped within the bonds of food
Animals use the sun’s energy indirectly through the food made by plants • Both plants and animals systematically break the bonds in food to make energy
ATP • Adenosine TriPhosphate • It is the major energy currency of the cell • It is made of a nitrogen base (adenine), a sugar (ribose), and three phosphate groups
When the third phosphate group of ATP is removed by hydrolysis, a large amount of energy is released. • Hydrolysis = when a molecule is broken down by adding water
ATP + H2O ADP + Pi • ADPis Adenosine DiPhosphate. • Pi is inorganic phosphate
Your body can use the energy released from breaking the 3rd phosphate bond of ATP to do work. • The amount of energy given off from ATP is the perfect amount for cells. • Your body can also make ATP from ADP.
CELLULAR RESPIRATION • Cellular respiration is the process by which the chemical energy of "food" molecules is partially transferred to ATP. • Breaking down food for energy.
Lipids, proteins, fats, and carbohydrates can be broken down for energy. • Our focus is on glucose.
Two types of respiration: • Aerobic– uses oxygen • Occurs in the mitochondria • Anaerobic – uses no oxygen (also called fermentation) • Occurs in the cytoplasm
AEROBIC RESPIRATON • The energy in the chemical bonds of glucose is partially transferred to ADP to makeATP. • 60% of the energy from glucose is “lost” as heat.
Glucose is broken down. • Oxygenis needed to completely break down glucose. • Carbon dioxide is released. Oxygen + GlucoseCO2 + water + energy
Glycolysis • The first set of reactions in cellular respiration is Glycolysis • Glycolysis is the process in which 1 molecule of glucose (C6) is broken in half, producing two molecules of pyruvic acid (C3) • Glycolysis occurs in the cytoplasm
Glucose ATP 2 2 ADP 4 ADP 2 NAD+ 4 ATP 2 NADH 2 Pyruvic Acid
ATP Production • Glycolysis is an energy-releasing process, but the cell needs to invest a little energy to get things started • 2 ATPs are needed to get glycolysis started, but at the end 4 ATPs are produced • Since we already invested 2 ATP, we have a net gain of 2 ATP
NADH Production • One of the reactions in glycolysis removes high energy electrons from each 3 carbon molecule and passes them to an electron carrier, NAD+ • NAD+ hold on to the electrons until it can pass them to other molecules within the mitochondria
The Krebs Cycle • After glycolysis, about 90% of the chemical energy of glucose is still trapped in the high-energy electrons of pyruvic acid • During the Krebs cycle, pyruvic acid is broken down and carbon dioxide is released • The Krebs cycle occurs in the mitochondria
The Krebs cycle begins when pyruvic acid enters the mitochondrion and is changed to another molecule • Pyruvic acid joins with a compound called Coenzyme-A to make acetyl-CoA • CO2 is given off • Acetyl-CoA joins with a 4 carbon molecule to make citric acid (6C)
Pyruvic Acid NAD+ CO2 NADH Co-A Co-A Co-A Citrate
Citric acid enters the Krebs cycle and is eventually broken down into a 4 carbon molecule • The products of the Krebs cycle include CO2, ATP, NADH, and FADH2 (a different electron carrier) • The Krebs cycle is where the energy pay-off is the largest
Citric Acid NAD+ NADH CO2 FAD NAD+ FADH2 NADH 4 Carbon Compound 5 Carbon Compound ADP NAD+ CO2 ATP NADH
What Happens to all of these Products??? • NADH and FADH2 go to the Electron Transport Chain to make ATPs • The CO2 released is the carbon dioxide you breathe out when you exhale • The ATPs can be used for cellular activity
Electron Transport • The Electron Transport Chain (ETC) is a series of proteins in the inner mitochondrial membrane that pass along the high energy electrons of NADH and FADH2 • These high-energy electrons are used by the ETC to convert ADP to ATP
How ATPs are Made • NADH and FADH2 bring their high energy electrons to specific proteins within the inner mitochondrial membrane • These special proteins are arranged in a line and make up the Electron Transport Chain
What’s the Point? • At the end of every ETC there is a special enzyme called ATP synthase • The H+ diffuse through the ATP synthase creating enough energy to add a phosphate to ADP to make ATP
What about our electrons? • At the end of the ETC the electrons need a place to go • Oxygen is the final electron acceptor which accepts two electrons and 2 H+ per oxygen atom • The point? No oxygen, no Krebs cycle
This oxygen is the oxygen that we breathe in every time we take a breath
WHOA! • This means that for each molecule of glucose you can make a maximum of 38 ATPs
ANAEROBIC RESPIRATION (FERMENTATION) • In many cells, if oxygen isnotpresent, glucose is broken down in a process called fermentation.
Less ATP is made in anaerobic respiration than in aerobic respiration. • In animals, lactic acid is made. • In yeast, alcohol (ethanol) and carbon dioxide are made.
Lactic Acid Fermentation • In animals, if enough oxygen is not present, respiration stops at Glycolysis • Pyruvic acid is converted to Lactic acid so that NAD+ can be regenerated
Lactic acid Glucose Pyruvic acid
ALCOHOLIC FERMENTATION • In yeast, if oxygen is not present, respiration also stops at Glycolysis • Pyruvic acid is converted to an Alcohol (ethanol) so that NAD+ can be regenerated
Pyruvic acid Glucose Ethanol
Aerobic vs. Anaerobic Respiration • Aerobic respiration gives us 38 ATPs (maximum) from each molecule of glucose • Anaerobic respiration gives us 2 ATPs from each molecule of glucose