Bonding
Bonding. By Mary Agarwala. Chemical Bonds?. What are they? Why do atoms form them? Why are some stronger than others?. Ionic Bonding. + charged ion is attracted to a – charged ion A nonmetal + a metal = ionic bond Ionic compound is entirely made up of ions

Bonding
E N D
Presentation Transcript
Bonding By Mary Agarwala
Chemical Bonds? • What are they? • Why do atoms form them? • Why are some stronger than others?
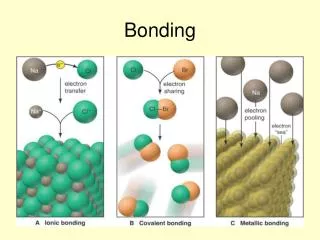
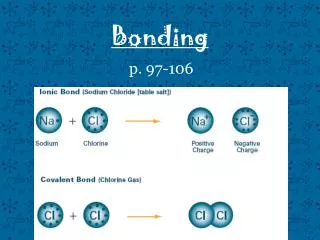
Ionic Bonding • + charged ion is attracted to a – charged ion • A nonmetal + a metal = ionic bond • Ionic compound is entirely made up of ions • Positively charged ion (Cation) • Negatively charged ion (Anion) • Ex. NaCl
Ionic properties • High melting points which means it ionic bonds are strong bond • Brittle • Dissolves in water, a process that breaks the ionic bonds and separates the ions (makes solutions that conduct electricity) • Polar…”Likes dissolves likes” • Liquid (motlen) ionic compounds conduct electricity • Solid ionic compounds do not conduct electricity well
OCTET RULE Atoms tend to gain, lose, or share electrons in order to acquire a full set of valence electrons
Lewis Dot Diagrams Valence electrons are represented as dots placed around the element symbol KNOW HOW TO WRITE IONIC COMPOUNDS
Empirical formula vs. Molecular formula Empirical Formula Molecular Formula Molecular compound Describes the composition of a molecular compound Tells how many atoms are in a single molecule of the compound Ex. Glucose (C6H12O6) • Denote the ratio of ions in a compound • Chemical formula • The lowest possible whole number subscripts for the elements • The compound wants to be electrically neutral
Covalent Bonds -A covalent bond is formed by a shared pair of electrons between two atoms -Each atom wants to be “octet happy” -Nonmetal + nonmetal = Covalent bond -This is best shown by combining atoms’ Lewis structures
Multiple Bonds Double Bonds Triple Bonds (show picture) (text book pg. 239 Eq.5) • (show picture) • (text book pg 239. Eq.4)
Exceptions to the Octet Rule • Atoms with less than an octet (BF3) • Atoms with more than an octet (SF4) • Molecules with an odd number of electrons (NO) • Don’t sweat the exceptions for the regents
Electronegativity • Property of an element that indicates how strongly an atom of that element attracts electrons in a chemical bond • When one atom is SIGNIFICANTLY MORE EN than another, the covalent bond is POLAR. • 2 atoms, in a bond, with SIMILAR ENs are NONPOLAR • KNOW EN TRENDS!!! Remember F is the biggest (EN=4.0)
Bond Type by Electronegativity Electronegativity Difference Bond Type Nonpolar covalent Polar covalent ionic • Less than or equal to 0.4 • Between 0.4 and 2.0 • Greater than or equal to 2.0
Naming Chemical Compounds Chemists name a compound according to the atoms and bonds that compose it
Naming Ionic Compounds • Write cation first • No need to specify number of atoms (AKA no prefixes) Ex. KI (potassium iodide) Ex. KNO3 (potassium nitrate) Ex. CuSO4 (copper(1) oxide)
Naming Molecular Compounds • Know numerical prefixes • Suffix –ide is added to the atom with a greater EN • Ex. NO2 (nitrogen dioxide) • Ex. BF3 (boron trifluoride) • Ex. P2O5 (diphosphoruspentoxide)
THE END “Good Luck.. Do Well!!!!”