STAT3
STAT3. Michael Patel. Overview. STAT3 is a transcription activator It homodimerizes and binds to the DNA promoter region It is associated with oncogenesis, cellular differentiation, and embryogenesis. STAT3 Signaling Pathway. Involved in signal transduction pathway


STAT3
E N D
Presentation Transcript
STAT3 Michael Patel
Overview • STAT3 is a transcription activator • It homodimerizes and binds to the DNA promoter region • It is associated with oncogenesis, cellular differentiation, and embryogenesis
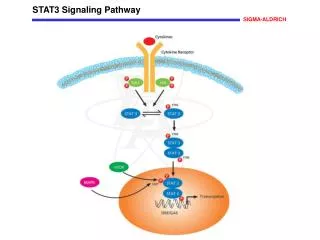
STAT3 Signaling Pathway • Involved in signal transduction pathway • Signal transduction translates external signals into internal cellular responses
Pathway Components • Cytokines are of the IL-6 (Interleukin 6) family, or very similar to them. • They bind to Glycoprotein 130 (gp130), an important cytokine receptor. • After a gp130 conformational change, Janus kinases (JAK) phosphorylate a STAT3 tyrosine, initiating STAT3 dimerization.
Pathway Components (cont.) • MAP and mTor are serine kinases, which phosphorylate S727. • This phosphorylation enhances DNA binding, although it is not essential.
Structure • STAT3 has three domains: Alpha, Binding, and SH2. • The Binding domain is responsible for binding to DNA. • SH2 is responsible for interacting with the tyrosine kinase.
Structure (cont.) PDB ID: 1bg1
SH2 Domain • Includes tyrosine (Y705) that, when phosphorylated, induces dimerization. • The polypeptide strand from residue 702-709 interacts in trans with the other monomer to form the dimer
Phosphotyrosine 705 Y705 shown in blue, rest of binding domain shown surrounding it. PDB ID: 1bg1
Phosphotyrosine 705 (cont.) Becker, et al. Nature 394, 145-151(9 July 1998)
DNA Binding • STAT3 binds to 9 base sequence: 5’-TTCCCGTAA -3’ • However, there may be a high sequence variability: 5’ – TTYN3YAA – 3’ (Y = T or C) • Four loops per monomer are in contact with DNA. (cx not pictured in following slide) • Bases -1, 0, and +1 can be variable because of water-mediated contact.
DNA Binding (cont.) Red = Polar Interactions Blue = Hydrophobic Interactions Becker, et al. Nature 394, 145-151(9 July 1998)
DNA Binding (cont.) Becker, et al. Nature 394, 145-151(9 July 1998)
DNA Binding (cont.) Complete STAT3 dimer bound to DNA. Becker, et al. Nature 394, 145-151(9 July 1998)
Regulation and p53 • In cancer cells, high amounts of active STAT3 are correlated with a mutant version of p53. • wt versions of p53 are incident with a lack of STAT3 dimerization • Thus, p53 or a downstream phosphatase dependent upon p53 acts as a tyrosine phosphatase, inhibiting dimerization.
Pias3 • Protein inhibitor of activated Stat3 • Binding domain is from residues 82-132 • Within this domain, the Y94 residue is essential for Pias binding. • Blocks STAT3 DNA binding.
Pias3 A is the lowest energy conformation. Levy, et al. Blood, 1 April 2006, Vol. 107, No. 7, pp. 2839-2845.
Rac1 • Conversely, Rac1 can activate increased amounts of STAT3. • It is associated with increased tyrosine and serine phosphorylation. • Mutation of phenylalanine (F37A) to alanine eliminates binding to STAT3. • Rac1 helps to recruit kinases to STAT3
Rac1 (cont.) Shown are GNP and phenylalanine (red) PDB ID: 1mh1
Oncogenesis • Persistently activated STAT3 is common among many epithelial cell cancers. • Can be the cause of over active kinases or lack of a proper inhibitor. • It is known to have anti-apototic effects. • Specifically, it activates the genes for such apoptosis inhibitors as Bcl-xL, c-Myc, cyclin D1, Vegf, and Survivin.
Embryogenesis • STAT3 has been found in mice to be essential for embryogenesis. • Specifically, it is required for embryo implantation in the uterus. • Additionally, deficiencies in pSTAT3 and IL-11, a cytokine, has been found in infertile women. Demiriadis, et al. Reproductive Biology and Endocrinology 2007, 5:44
Possible Treatment Directions • Targeted deletion in skin cells prevents cancer. • One possible treatment involved the injection of antisense oligonucleotides that have no effect on healthy cells. • Platinum containing compounds can disrupt STAT signaling. • Another possibility is use of inhibitors that interact with the tyrosine kinases.