Chapter 5 STEREOCHEMISTRY
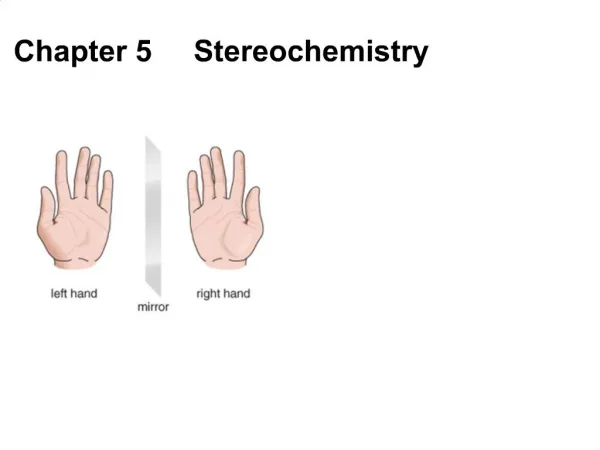
Chapter 5 STEREOCHEMISTRY. Stereochemistry. CHIRALITY AND ENATIOMERS. Chiral Object – an object that IS NOT superimposable with its mirror image. Achiral Object – an object that IS superimposable (i.e. identical) with its mirror image.


Chapter 5 STEREOCHEMISTRY
E N D
Presentation Transcript
Chapter 5STEREOCHEMISTRY Stereochemistry
CHIRALITY AND ENATIOMERS • Chiral Object – an object that IS NOT superimposable with its mirror image. • Achiral Object – an object that IS superimposable (i.e. identical) with its mirror image. • Enantiomers – stereoisomers, which are non-superimposable mirror images. Stereochemistry
CHIRAL CARBON ATOMS • Chiral Carbon Atoms (also known as asymmetric carbon atoms) – carbon atoms bonded to FOUR DIFFERENT groups. Chiral atoms are generally called chiral centers. * * mirror • Some rules: • If a molecule has NO chiral center, it is usually, but not necessarily achiral. • If a molecule has just ONE chiral center, it is definitely chiral. • If a molecule has MORE THAN ONE chiral center, it may or may not be chiral. Stereochemistry
THE MIRROR IMAGES OF CARBON CENTERS WITH THREE DIFFERENT SUBSTITUENTS ARE SUPERIMPOSABLE 120o mirror IDENTICAL WITH THE ORIGINAL Stereochemistry
THE MIRROR IMAGES OF CARBON CENTERS WITH FOUR DIFFERENT SUBSTITUENTS (CHIRAL CARBON ATOMS) ARE NOT SUPERIMPOSABLE 120o * * * mirror NOT IDENTICAL WITH THE ORIGINAL Stereochemistry
CARBON CENTERS WITH FOUR DIFFERENT SUBSTITTUENTS (CHIRAL CARBON ATOMS) DO NOT HAVE INTERNAL MIRROR PLANES OF SYMMETRY Stereochemistry
CHIRAL COMPOUNDS ALSO DO NOT HAVE INTERNAL MIRROR PLANES OF SYMMETRY MIRROR IMAGE IDENTICAL WITH ORIGINAL MIRROR IMAGE IS NOT IDENTICAL WITH ORIGINAL Stereochemistry
THE CAHN – INGOLD – PRELOG CONVENTION • Assign a “priority” and a number to each group, bonded to the chiral carbon atom. Highest priority group gets number 1, second in priority – number 2, third in priority – number 3, lowest in priority – number 4. • First compare the atoms immediately bonded to the chiral center. The atom with highest atomic weight is highest in priority and therefore # 1, the atom with lowest atomic weight is #4. • If two or more atoms of the immediate environment are identical, compare the next set of atoms along the chains, applying the criterion from (a). • REMEMBER: One higher-priority atom is more important • than any number of lower-priority atoms. Stereochemistry
THE CAHN – INGOLD – PRELOG CONVENTION • Treat double- and triple-bonded atoms as an equivalent number of single-bonded atoms, i.e. imagine that the p-bonds are broken and the atoms at both ends of the bond are duplicated (if it is a double bond) or triplicated (if it is a triple bond). Stereochemistry
THE CAHN – INGOLD – PRELOG CONVENTION 2. Orient the molecule in a fashion such that the lowest in priority group (number 4) is in back and view the molecule along the bond from the chiral center to group #4. Draw a curved arrow, starting from #1, through #2, to #3. If the arrow points in a clockwise direction – R-enantiomer, if the arrow points in a counter-clockwise direction – S-enantiomer. Stereochemistry
THALIDOMIDE: DISASTROUS BIOLOGICAL ACTIVITY OF THE “WRONG” ENANTIOMER Stereochemistry