Energy and Reactivity in Respiration Cascade Reactions
60 likes | 135 Vues
Explore the chemical reactions in the respiratory cascade, from Complex I to Proton-translocating ATPase, highlighting energy changes, electron transfer processes, and proton gradients in physical chemistry. Witness how molecules collide and react to facilitate cellular energy production.

Energy and Reactivity in Respiration Cascade Reactions
E N D
Presentation Transcript
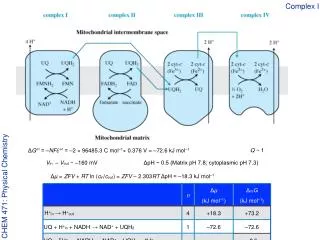
Complex I ΔG°′ = –NFℰ°′ = –2 × 96485.3 C mol–1× 0.376 V = –72.6 kJ mol–1 Q ~ 1 CHEM 471: Physical Chemistry Vin – Vout ~ –160 mV ΔpH ~ 0.5 (Matrix pH 7.8; cytoplasmic pH 7.3) Δµ = ZFV + RT ln (cin/cout) = ZFV – 2.303RT ΔpH = –18.3 kJ mol–1
The respiratory cascade CHEM 471: Physical Chemistry NAD+ + H+in + 2e– → NADHℰ°′ = –0.324 V UQ + 2H+in + 2e– → UQH2ℰ°′ = +0.052 V Reversing the first half-reaction, and adding, the overall reaction is strongly favorable UQ + H+in + NADH→ NAD+ + UQH2ℰ°′ = +0.376 V ΔG°′ = –NFℰ°′ = –2 × 96485.3 C mol–1× 0.376 V = –72.6 kJ mol–1
Complex III UQ + 2H+in + 2e– → UQH2ℰ°′ = +0.052 V cyt-c(Fe3+) + e– → cyt-c(Fe2+)ℰ°′ = +0.254 V CHEM 471: Physical Chemistry 2 cyt-c(Fe3+) + UQH2→ 2 cyt-c(Fe2+) + UQ + 2 H+outℰ°′ = +0.202 V ΔG°′ = –NFℰ°′ = –2 × 96485.3 C mol–1× 0.202 V = –39.0 kJ mol–1
Complex IV cyt-c(Fe3+) + e– → cyt-c(Fe2+)ℰ°′ = +0.254 V ½O2 + 2H+ + 2e– → H2Oℰ°′ = +0.815 V CHEM 471: Physical Chemistry ½O2 + 2H+ + 2 cyt-c(Fe2+) → H2O + 2 cyt-c(Fe3+)ℰ°′ = +0.561 V ΔG°′ = –NFℰ°′ = –2 × 96485.3 C mol–1× 0.561 V = –109.3 kJ mol–1
Proton translocating ATPase CHEM 471: Physical Chemistry
The collision tube hit! d hit! miss! Area of tube = σ = πd2 CHEM 471: Physical Chemistry In a time dt the molecule travels <s>dt. Volume of tube dV = σ<s>dt. Number of molecules per unit volume of the tube dN/dV = ρN. ⇒ dN = ρNdV= ρNσ<s>dt. Each molecule in the tube results in a collision. Number of collisions per unit time = z= dN/dt = ρNσ<s>