Unit VII Crystal structure
Unit VII Crystal structure. Dr. Ravindra H.J & Dr. P.S. Aithal. Objectives. Definition of Space lattice, Bravais lattice, Unit cell, Primitive cell and Lattice parameters Seven crystal systems and 14 Bravais lattices Direction and planes in a crystal, Miller indices and interplanar spacing

Unit VII Crystal structure
E N D
Presentation Transcript
Unit VIICrystal structure • Dr. Ravindra H.J & Dr. P.S. Aithal
Objectives • Definition of Space lattice, Bravais lattice, Unit cell, Primitive cell and Lattice parameters • Seven crystal systems and 14 Bravais lattices • Direction and planes in a crystal, Miller indices and interplanar spacing • Co-ordination number of cubic structure, Number of atoms per unit cell, Relation between Atomic radius and Lattice constant, and Atomic packing factor • Bragg’s Law and Bragg’s x-ray spectrometer • Crystal structures of NaCl and diamond.
Introduction TO CRYSTALS • Gas • Liquids • Solids - crystals Amorphous Crystal: The solids which possess the long range, three dimensional ordering are known as crystals. Definition: a solid which possesses long range, three dimensional atomic or molecular arrangements
Motif Lattice points Space lattice • A crystal structure is built of infinite repetition of identical atoms or group of atoms in three dimensions. • A group of atoms is called the basis or motif. • The set of mathematical points to which the basis is attached is called a lattice.
Space lattice • A crystal lattice or space latticeis an infinite pattern of lattice points in three dimensions, each of which must have the same surroundings in the same orientation. OR Space lattice is a well defined repeating unit (motif or basis) and the each motif is associated to a lattice point so that the overall lattice is a three dimensional array of lattice points which are geometrically identical. Space lattice = Lattice * Motif (Basis) here the symbol * refers to mean “associated with” In simple metallic crystals the number of atoms allocated to each lattice point is just one and in the case of a protein crystal the number of atoms allocated to each lattice point may be thousands.
Motif Bravais lattice and non-Bravais lattice • The array of equivalent lattice points in two or in three dimensions is referred as Bravais lattice. • The array of non-equivalent lattice points is referred as non-Bravais lattice.
D C B A Basis vector and translational vector • A vector is a mathematical quantity having both magnitude and direction. • A vector having unit magnitude is generally referred as a unit vector. • The vectors which gives the magnitude and direction of separation between two nearest lattice points is called basis vector.
D C B A Translational vector or lattice vector • If we chose three basis vectors, , and to define a right-handed, orthogonal, three dimensional Cartesian co-ordinate systems then any vector T which gives the position and direction of the any identical lattice point in three dimensional lattices can be represented as • Where, is the translational vector or lattice vector and u, v, and w are integers. The coordinates of the lattice point in three dimension is represented by (u,v,w). E
Unit cell • The parallelepiped-shaped volume which, when reproduced by close packing in three dimensions, gives the whole crystal structure or space lattice itself is called the unit cell. • Definition: Unit cell is the smallest geometrical volume in a space lattice, defined by three basis vectors, which on translation along three basis vector directions yields the complete crystal structure or space lattice itself.
Primitive cell and non-primitive cell • All fundamental unit cells associated with a single lattice point in it are called primitive cells or P-cells. • A primitive cell is a one in which lattice points are present only at the edges (or corners) of the unit cell. • The unit cells associated with more than one lattice point are referred as non-primitive or multi unit cells. • A non-primitive cell is a one in which there are lattice points present in the unit cell in addition to the lattice points at the edges of the unit cell.
Lattice parameters • Unit cell parameters is defined as a set of six parameters, lengths of the three non-collinear basis vectors and three interfacial angles, that together defines a unit cell. • Basically the parallelepiped formed by the three basis vectors , and , defines the unit cell of the lattice, with edges of length a, b and c, is selected as unit cell.
Crystal systems & Bravais lattices • The systematic work of describing the space lattices was done initially by Frankenheim, who, in 1835, proposed that there were fifteen space lattices. • Bravais In 1848 proposed there are only 14 space lattices • These 14 Bravais lattices fall into seven crystal systems • Cubic • Tetragonal • Orthorhombic • Monoclinic • Triclinic • Rhombohedra (or Trigonal) and • Hexagonal.
Polymorphism • Ability of any substance to crystallize in several solid phases that possess different lattice structures. • Example : Fe – BCC at room temperature • At 9100C BCC to FCC • >14000C FCC to BCC • Type of the lattice also affects the properties of the Iron.
Directions and planes in a crystal lattice • Any lattice point T can be represented by the equation • The direction in space corresponding to the vectors is written in square brackets, without using commas to separate the digits, as [uvw]. • For example [213] and it should be read as ‘two one three’.
Planes in a crystal and Miller indices • A set of three integers which are used to represent the plane are referred as Miller integers. • A set of planes is defined by three integers h,k, and l , known as plane indices or Miller indices. Procedure to find Miller indices • Find the intercept of the planes along three basis vectors. • Either reduce the fractional intercepts to set of integers p, q • and r or express the intercepts as fractional multiples of • basis vector lengths • Take the reciprocals • Find the least common multiple of the denominator and • multiply this LCM to above ratios yields set of three integers • h, k and l referred as Miller indices.
Coordination number • The coordination number is defined as the number of nearest neighbors surrounding the central or a given atom within the space lattice. • Simple cubic- coordination number is 6 • Body centered cubic – coordination number is 8 • Face centered cubic – coordination number is 12 • Number of atoms per unit cell • Simple cubic - (1/8 X 8) = 1 • Body centered cubic - (1/8 X 8) +1 =2 • Face centered cubic - (1/8 X 8 ) +(1/2 X 6) =4
Relation between Atomic radius and Lattice constant • Simple cubic: a = 2R • Body centered cubic • Face centered cubic
Atomic packing factor (APF) • Atomic packing factor is the ratio of the total volume of the atoms present in one unit cell to that of total volume of the unit cell. • APF = volume of the number of atoms present in a unit cell/ volume of the unit cell • SC • BCC FCC
Expression for a space lattice constant “a” for a cubic lattice The density of the unit cell is the ratio of total mass of the atoms or molecules belonging to unit cell to that of volume of unit cell
Crystal structure of NaCl • Type of the lattice is determined by considering only one type of atom • NaCl is an ionic compound • Coordination number is 6 • Distance between two same type ion is 5.62 Å • Number of NaCl molecules per unit cell is 4 • Number of atoms per unit cell is 8
Calculation of lattice constant of NaCl • Atomic weight of Na is 23 • Atomic weight of Cl is 35.45 • Density of NaCl is 2180 kg/m3 • Number of NaCl molecules in a Unit cell is 4
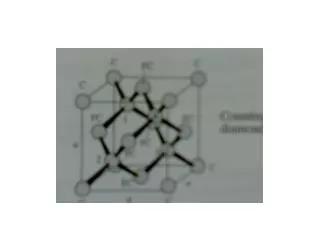
Crystal structure of diamond • The diamond structure can be described as two interpenetrating fcc structures that are displaced relative to one another along the main diagonal. • The four carbon atoms together forms tetrahedron structure • The nearest neighbor distance in the • diamond structure is • there are 8 atoms in a unit cell • packing factor Is or 0.34. • The elements which crystallize similar to the structure of diamond are Germanium (Ge) and Silicon (Si).
Problems • Draw the following planes in a cubic cell [312] • A monochromatic X-ray beam of wavelength 1.5Å undergoes second order Bragg reflection from the plane (2 1 1) of cubic crystal at a glancing angle of 54.38o. Calculate the lattice constant. • Calculate the glancing angle for incidence of X-rays of wavelength 0.58 Å on the plane (1 3 2) of NaCl which results in second order diffraction maxima taking the lattice as 3.81 Å. • The minimum order of Bragg’s reflection occurs at an angle of 20o in the plane [212]. Find the wavelength of X-ray if lattice constant is 3.615 Å. • In a cubic unit cell the distance between the two atoms present at the coordinates (100) and (011) is 5.8 Å. A monochromatic electron beam of unknown wavelength undergoes second order Bragg reflection from the plane (025) at a glancing angle of 45.95o. Calculate the wavelength of the electron beam.