Epithelial Nitric Oxide Synthase (eNOS)
Epithelial Nitric Oxide Synthase (eNOS). SIGMA-ALDRICH. Epithelial Nitric Oxide Synthase (eNOS)

Epithelial Nitric Oxide Synthase (eNOS)
E N D
Presentation Transcript
Epithelial Nitric Oxide Synthase (eNOS) SIGMA-ALDRICH
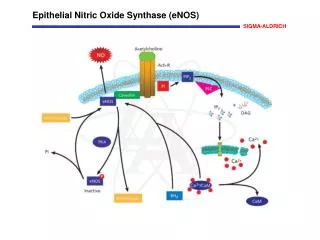
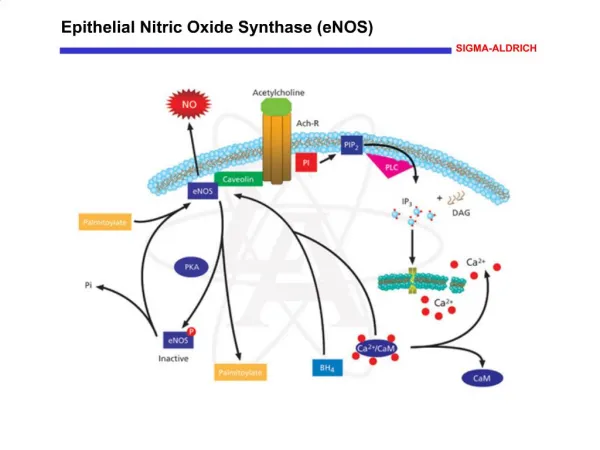
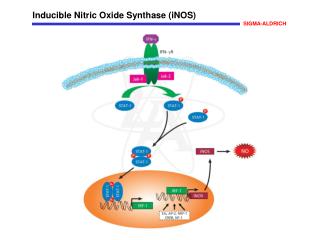
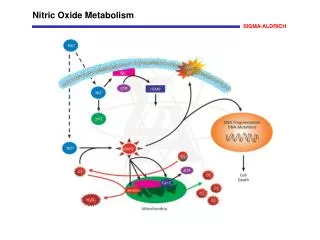
Epithelial Nitric Oxide Synthase (eNOS) Nitric Oxide (NO) produced in the endothelial cells is involved in vasorelaxation, platelet aggregation, and mechanisms of cardiovascular homeostasis. Endothelial nitric oxide synthase (eNOS, cNOS, Type III) is constitutively expressed in endothelial and other cell types. Myristoylation and palmitoylation maintain the localization of eNOS to caveolae in the plasma membrane of resting cells where it is bound to caveolin. eNOS is inactive in the membrane-bound state. Activation of endothelial acetylcholine receptors activate phospholipase C (PLC) that catalyzes the production of inositol 1,4,5-trisphosphate (IP3) and diacylglycerol (DAG) from phosphatidylinositol 4,5-bisphosphate (PIP2). The IP3-induced increase in intracellular Ca2+ activates calmodulin that binds to eNOS, which dissociates from caveolin and translocates to the cytoplasm. Phosphorylation of eNOS by protein kinase A (PKA) inactivates the enzyme, which then relocates to the membrane caveoli. References Ghosh, S., et al., Interaction between caveolin-1 and the reductase domain of endothelial nitric-oxide synthase. Consequences for catalysis. J. Biol. Chem., 273, 22267-22271 (1998). Arnal, J.F., et al., Endothelium-derived nitric oxide and vascular physiology and pathology. Cell. Mol. Life Sci., 55, 1078-1087 (1999). Yeh, D.C., et al., Depalmitoylation of endothelial nitric-oxide synthase by acylprotein thioesterase 1 is potentiated by Ca2+-calmodulin. J. Biol. Chem., 274, 33148-33154 (1999).