Inhaled Nitric Oxide
Inhaled Nitric Oxide. A study on the effects of inhaled Nitric Oxide (iNO) in neonatal Pulmonary Hypertension . by: Cecilia Cherian Marc Chiappetta Jay Suerte Santos Machoka. Biological Question.

Inhaled Nitric Oxide
E N D
Presentation Transcript
A study on the effects of inhaled Nitric Oxide (iNO) in neonatal Pulmonary Hypertension by: • Cecilia Cherian • Marc Chiappetta • Jay Suerte • Santos Machoka
Biological Question • Is the administration of low dose versus high dose nitric oxide effective in the treatment of persistent pulmonary hypertension of newborn ?
Hypothesis • Administering low dose inhaled nitric oxide improves oxygen delivery and compliance, thus is beneficial in treating persistent pulmonary hypertension of newborns.
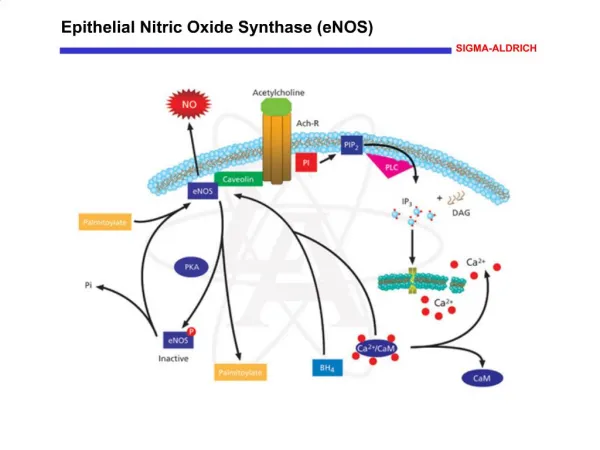
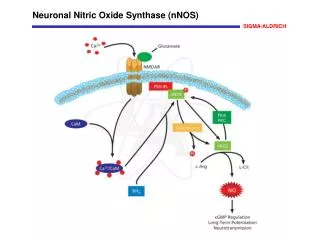
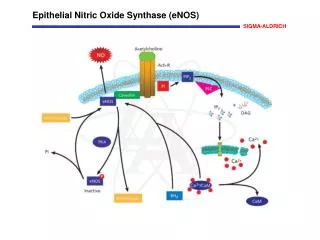
What is Nitric Oxide ? • Nitric Oxide is a highly diffusible, lipid soluble free radical that oxidizes quickly to nitrogen dioxide (NO2) in the presence of oxygen • It is produced internally in every cell and organ in the human body.
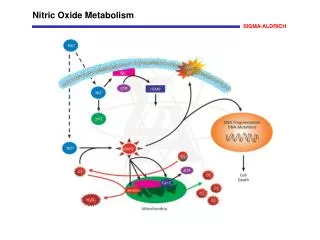
How iNO works • iNO is a selective pulmonary vasodilator. • iNO will dilate only the pulmonary blood vessels adjacent to functioning alveoli. • iNO diffuse into the capillaries, free NO immediately binds to hemoglobin forming nitrosylhemoglobin, which is rapidly oxidized to methmoglobin and eventually undergoes conversion to reduced the hemoglobin
Indications for iNO • iNO is used to treat pulmonary hypertension such as primary pulmonary hypertenison(PPH) and persistent pulmonary hypertension of newborns (PPHN). • Neonates with hypoxic respiratory failure associated pulmonary hypertension. • NO therapy is often used with other forms of therapy including high frequency oscillatory ventilation and surfactant therapy, both of which are aimed at improving oxygen delivery and lung compliance.
Dosage • Initial dosage was 20 ppm(particles per million). • Often reduced to 5 ppm at the end of 4 hours of initial treatment. • Reduce inhaled nitric oxide dose by 50% (20, 10,5, 1 ppm).
Methods of administration • NO is delivered with the O2 at a constant concentration throughout the breathing cycle. • Set NO level is stable over a wide range of flows and flow patterns, including spontaneous breathing modes. • The flow sensor placed in the outlet of the ventilator upstream from the humidifier measures minute ventilation (lpm).
Method of administration • The injector varied the nitric oxide based on the measured ventilation. • Nitric oxide, nitrogen dioxide and oxygen are measured in the inspiratory limb of the circuit, just before the patient wye and a calibration mode verified proper analyzer function. High and low alarms are provided for all three gases and an automatic cut-off to prevent nitric oxide overdose.
Hazards and contraindications • Nitric oxide when combined with oxygen produces nitrogen dioxide (NO2), which is a toxic gas. • Because of it’s very minimal half life (0.1 seconds – 5 seconds), it is quickly inactivated once it combines with hemoglobin. • Although rare, the patients as well as health care providers can be adversely affected. • Factors influencing NO2 production are O2 concentration, NO concentration and time of contact between NO and O2.
Hazards and contraindications • Patients most at risk include those receiving high oxygen concentration and low ventilator flow rates. • Production of methemoglobin can be a problem due to anemic hypoxia which can be treated with vitamin C.. • A major contraindications of this therapy is children who as a result of congenital cardiac anomalies need a right-to-left shunt to survive and NO therapy corrects the right-to-left shunts.
methodology • A patient group of 34 weeks gestation to 8 days old were selected for the iNO treatment. • All these patients had persistent pulmonary hypertension and severe respiratory failure requiring mechanical ventilation. • Infants that were diagnosed with pneumonia, sepsis, and meconium aspiration were included. • Infants diagnosed with congestive heart defect (CHD) were excluded because previous studies showed that these infants did not respond to iNO therapy.
methodology • Data collection • After parental consent was obtained, eligible patients with 2 separate Oxygen Indices (OIs) of >10 were randomized to receive a starting iNO dose of either 1 to 2 ppm (low dose group) or 10 to 20 (high dose group). • In the high dose group the therapy was started at 20 ppm and could be decreased to 5 ppm in the first 24 hours. • In the low dose group starting at 1 ppm and not obtaining a positive result, increasing the ppm in small increments were made until desired results were obtained. (increased oxygen index)
methodology • Data collection • The response of iNO dose was assessed according to the increase in arterial PaO2 and decrease in OI 30 to 60 minutes after exposure to initial starting concentration. • After the first 24 hrs. they had to be weaned to 5 ppm for no more than 96 hours.
methodology • Data collection • Failure to improve after 96 hours were considered for ECMO therapy. • All discontinuation of therapy was performed at doses of 0.5 to 1 ppm. • When any response occurred to 40 ppm or greater, attempts were made every 4 hours to halve the dose, until the infant was receiving 20 ppm.
methodology • Data collection • During the treatment period no changes were made to ventilator or any other therapies. • Methemoglobin analysis was performed before iNO administration and at 6 hours and every 8 hours during iNO administration.
Statistics • An objective discussion of the three research papers clinical findings indicated the validity of inhaled nitric oxide on the neonatal population.
In the neonatal population that ended up needing ECMO • Of the group that initially had nitric oxide therapy 47% of them did survive the hospital stay, while only 36 % of the control group survived (where N= 248+38=286 patients) • These patient population was identical in gestation (mean 35 wks old), age (mean 2.5 days old), the control group started with an initial treatment of 0ppm iNO, and an oxygenation index of >25.
Conclusion • While one paper proves objectively that starting patients at low doses of nitric oxide can have an inhibiting effect on future increments of nitric oxide therapy all the papers agree that nitric oxide therapy is more useful than 100 % oxygen in the treatment of PPHN secondary to pulmonary hypoxia.
Conclusion • Gradually weaning of the nitric oxide to an acceptable dose is carefully monitored by using different criteria including but not limited to oxygen indices and ABGs. • Nitric oxide is used in conjunction with oscillator therapy.
Conclusion • Nitric Oxide is used as a precursor to ECMO therapy which is more expensive and has more risk factors.
Glossary • 1. Persistent Pulmonary Hypertension ofNewborn (PPHN)- A syndrome that causes right to left shunt, severe hypoxemia, and mixed acidosis. • 2. Parts Per Million (ppm)- Unit of measurement used for nitric oxide. • 3. Nitric Oxide (NO)- A selective pulmonary vasodilator that improves blood flow to ventilated alveoli, resulting decreased shunt and improve oxygenation.
Glossary • 4. Nitrogen Dioxide (NO2)- A toxic gas produced when nitric oxide is combined with oxygen. • 5. Methemoglobin (metHb)- The result when Nitric Oxide combines with Hemoglobin, resulting in oxygen desaturations. • 6. Hemoglobin (Hb)- Oxygen carrying protein found in the blood that carries oxygen from the lungs to the cells.
Reference • 1. Clark, R.H., Kueser, T.J., & Walker, M.W. Low-DoseNitric Oxide Therapy for Persistent Pulmonary Hypertension of the Newborn. The New England Journal of Medicine. (Feb. 17, 2000). Retrieved from: http://content.nejm.org/cgi/content/full/342/7/469?ijkey=a3f529d65be4b34d2f21ab8cb3b. • 2. Finer, N.N., Sun, J.W., Rich, W. Randomized, Prospective Study of Low Dose Versus High Dose Inhaled Nitric Oxide in the Neonate with HypoxicRespiratory Failure. Journal of the Academy of Pediatrics. (Oct. 2001). Retrieved from: http://pediatrics.aappublications.org/cgi/content/full/108/4/949. • 3. Cornfield, D.N., Maynard, R.C., & Deregnier, R. Randomized , Controlled Trial of Low Dose Inhaled Nitric Oxide in the Treatment of Term and Near Term Infants with Respiratory Failure and Pulmonary Hypertension. Official Journal of the American Academy of Pediatrics. (Nov. 1999). Retrieved from: http://pediatrics.aappublications.org/cgi/content/full/105/5/1089
Extra links • How to set up a nitric oxide system