Chapter 2 Matter and Change
Chapter 2 Matter and Change. Section 2.1 Matter. OBJECTIVES: Identify the characteristics of matter and substances. Differentiate among the three states of matter. Define physical property, and list several common physical properties of substances. What is Matter?.

Chapter 2 Matter and Change
E N D
Presentation Transcript
Section 2.1 Matter • OBJECTIVES: • Identify the characteristics of matter and substances. • Differentiate among the three states of matter. • Define physical property, and list several common physical properties of substances.
What is Matter? • Matter is anything that takes up space and has mass. • Mass- amount of material or “stuff” in an object • Weight is due to gravity, and changes from location to location; mass is always constant.
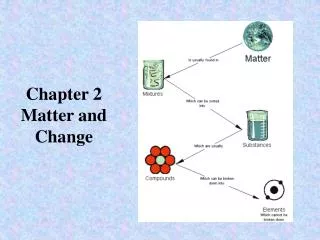
Types of Matter • Substance- a particular kind of matter - pure; is uniform (all the same) and has a definite composition (examples are elements & compounds) • water; gold • Mixture- more than one kind of matter; has a variable composition
Properties Physical Properties- a property that can be observed and measured without changing the composition. • Examples- color, hardness, m.p., b.p., malleability Chemical Properties- a property that can only be observed by changing the composition of the material • Flammability, burn, rot, rust, decompose, ferment, corrode, explode
States of matter • Solid- matter that can not flow (definite shape) and has definite volume. • Liquid- definite volume but takes the shape of its container (flows). • Gas- a substance without definite volume or shape and can flow. • Vapor- a substance that is currently a gas, but normally is a liquid or solid at room temperature. (water vapor?)
States of Matter Definite Volume? Definite Shape? Compressible? Solid YES YES NO Liquid NO NO YES Gas NO NO YES
Condense Freeze Evaporate Melt Gas Liquid Solid
Changes Physical changes -A change that changes appearances, without changing the composition. • Ex. Boil, melt, cut, bend, split, crack • Boiled water is still water. Chemical changes - a change where a new form of matter is formed. • Ex. Rust, burn, decompose, ferment
Section 2.2 Mixtures • OBJECTIVES: • Categorize a sample of matter as a substance or a mixture. • Distinguish between homogeneous and heterogeneous samples of matter.
Mixtures =physical blend of at least two substances; variable composition • Heterogeneous- mixture is not uniform in composition • Chocolate chip cookie, gravel, soil. • Homogeneous- same composition throughout; called “solutions” • salt water, cough syrup • Every part keeps it’s own properties.
Solutions =Homogeneous mixture • Mixed molecule by molecule • Can occur between any state of matter • Table 2.3, page 33 • gas in gas; liquid in gas; gas in liquid; solid in liquid; solid in solid (alloys), etc.
Solutions • Like all mixtures, they keep the properties of the components. • Some can be separated easily by physical means: rocks and marbles, iron filings and sulfur • Other methods: distillation- takes advantage of different boiling points
Section 2.3 Elements and Compounds • OBJECTIVES: • Explain the differences between an element and a compound. • Identify the chemical symbols of common elements, and name common elements given their symbols.
Substances • Elements- simplest kind of matter • cannot be broken down any simpler • all one kind of atom. • Compounds are substances that can be broken down only by chemical methods • When broken down, the pieces have completely different properties than the original compound. • Made of two or more atoms, chemically combined (not physical blend!)
Made of one kind of material Made of more than one kind of material Made by a chemical change Made by a physical change Definite composition Variable composition Compound or Mixture Compound Mixture
Element Compound or Mixture? Which is it?
Chemical Symbols & Formulas • Currently, there are 115 elements • Each has a 1 or two letter symbol • First letter always capitalized; the second never; chemical “shorthand” • Don’t need to memorize all of them; know Table A.3 p. 886 • Some from Latin or other languages; note Table 2.4, page 40
Section 2.4 Chemical Reactions • OBJECTIVES: • Differentiate between physical and chemical changes in matter. • Apply the Law of Conservation of Mass.
Chemical Reactions =When one or more substances are changed into new substances • Reactants- stuff you start with • Products- What you make • ability to undergo chemical reaction is called a chemical property • products have NEW PROPERTIES • Arrow from reactants to products
Chemical Reactions Potassium + Sulfur Potassium sulfide reactants products
Indications of a chemical reaction: • Energy absorbed or released (temperature changes hotter or colder) • Color change • Gas production (bubbling, fizzing, or odor change) • Formation of aprecipitate- a solid that separates from solution (won’t dissolve) • Irreversibility- not easily reversed
Law of Conservation of Mass =Mass can not be created or destroyed in ordinary (not nuclear) chemical reactions or physical change • Which means: • All the mass can be accounted for • Burning of wood results in products that appear to have less mass as ash; where is the rest?