Skeletal Muscle Physiology
390 likes | 755 Vues
Skeletal Muscle Physiology. Susan V. Brooks Herzog Department of Physiology University of Michigan. Structural hierarchy of skeletal muscle. Muscle. A little less than half of the body’s mass is composed of skeletal muscle, with most muscles linked to bones by tendons through which

Skeletal Muscle Physiology
E N D
Presentation Transcript
Skeletal Muscle Physiology Susan V. Brooks Herzog Department of Physiology University of Michigan
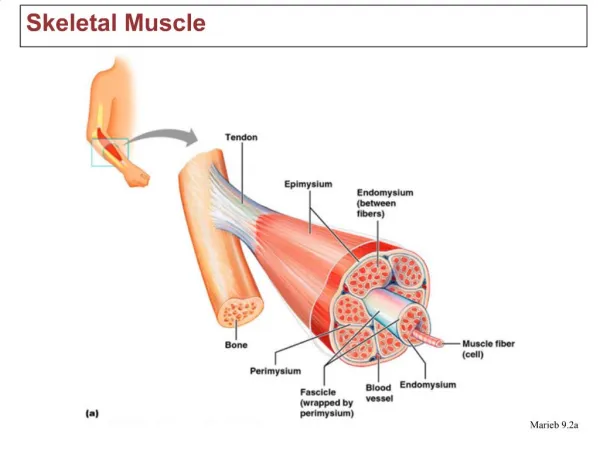
Structural hierarchy of skeletal muscle Muscle A little less than half of the body’s mass is composed of skeletal muscle, with most muscles linked to bones by tendons through which the forces and movements developed during contractions are transmitted to the skeleton. Muscle fibers Muscle fiber Myofibril Sarcomere Modified from McMahon, Muscles, Reflexes and Locomotion Princeton University Press, 1984.
Sarcomere: functional unit of striated muscle Cross-sectional views of: Myosin filaments Actin filaments Z line Actin filaments thin filament lattice overlap region center of sarcomere thick filament lattice Electron micrograph 1 mm I band I band A band Sarcomere Modified from Vander, Sherman, Luciano Human Physiology, McGraw-Hill.
Myosin is a molecular motor Myosin is a hexamer: 2 myosin heavy chains 4 myosin light chains Coiled coil of two a helices 2 nm C terminus Myosin head: retains all of the motor functions of myosin, i.e. the ability to produce movement and force. Nucleotide binding site Myosin S1 fragment crystal structure Ruegg et al., (2002) News Physiol Sci 17:213-218. NH2-terminal catalytic (motor) domain neck region/lever arm Modified from Vander, Sherman, Luciano Human Physiology, McGraw-Hill.
Hypothetical model of the swinging lever arm Working stroke produced by opening and closing of the nucleotide binding site, resulting in rotation of the regulatory domain (neck) about a fulcrum (converter domain). Sub-nanometer rearrangements at active site are geared up to give 5- 10 nm displacement at the end of the lever arm. Power Stroke Ruegg et al., (2002) News Physiol Sci 17:213-218.
How striated muscle works: The Sliding Filament Model From Vander, Sherman, Luciano Human Physiology, McGraw-Hill. The lever movement drives displacement of the actin filament relative to the myosin head (~5 nm), and by deforming internal elastic structures, produces force (~5 pN). Thick and thin filaments interdigitate and “slide” relative to each other.
Chemomechanical coupling – conversion of chemical energy (ATP about 7 kcal/mole) into force/movement. • ATP is unstable thermodynamically • Two most energetically favorable steps: • 1. ATP binding to myosin • 2. Phosphate release from myosin • Rate of cycling determined by M·ATPase activity and external load Adapted from Goldman & Brenner (1987) Ann Rev Physiol 49:629-636.
Shortening velocity dependent on ATPase activity Different myosin heavy chains (MHCs) have different ATPase activities. There are at least 7 separate skeletal muscle MHC genes…arranged in series on chromosome 17. Two cardiac MHC genes located in tandem on chromosome 14. The slow b cardiac MHC is the predominant gene expressed in slow fibers of mammals. Goldspink (1999) J Anat 194:323-334.
Power output: the most physiologically relevant marker of performance Power = work / time = force x distance / time = force x velocity Peak power obtained at intermediate loads and intermediate velocities. Figure from Berne and Levy, Physiology Mosby—Year Book, Inc., 1993.
Most likely to cause muscle injury Three potential actions during muscle contraction: (Isotonic: shortening against fixed load, speed dependent on M·ATPase activity and load) Biceps muscle shortens during contraction • shortening • isometric • lengthening Biceps muscle lengthens during contraction Modified from Vander, Sherman, Luciano Human Physiology, McGraw-Hill.
Spinal cord • To increase force: • Recruit more M.U.s • Increase freq. • (force –frequency) Motor Units: motor neuron and the muscle fibers it innervates • The smallest amount of muscle that can be activated voluntarily. • Gradation of force in skeletal • muscle is coordinated largely by the nervous system. • Recruitment of motor units • is the most important means • of controlling muscle tension. Modified from Vander, Sherman, Luciano Human Physiology, McGraw-Hill. • Since all fibers in the motor • unit contract simultaneously, • pressures for gene expression • (e.g. frequency of stimulation, • load) are identical in all fibers • of a motor unit. From Matthews GG Cellular Physiology of Nerve and Muscle Blackwell Scientific Publications.
Physiological profiles of motor units: all fibers in a motor unit are of the same fiber type • Slow motor units contain slow fibers: • Myosin with long cycle time and therefore uses ATP at a slow rate. • Many mitochondria, so large capacity to replenish ATP. • Economical maintenance of force during isometric contractions and efficient performance of repetitive slow isotonic contractions. • Fast motor units contain fast fibers: • Myosin with rapid cycling rates. • For higher power or when isometric force produced by slow motor units is insufficient. • Type 2A fibers are fast and adapted for producing sustained power. • Type 2X fibers are faster, but non-oxidative and fatigue rapidly. • 2X/2D not 2B. Modified from Burke and Tsairis, Ann NY Acad Sci 228:145-159, 1974.
Continuum of Physical Activity strength trained endurance trained Load controls inactivity Frequency of recruitment Muscle is plastic! Muscle “adapts” to meet the habitual level of demand placed on it, i.e. level of physical activity. Level of physical activity determined by the frequency of recruit- ment and the load. Increase muscle use – endurance training – strength training (cannot be optimally trained for both strength and endurance) Decrease muscle use – prolonged bed rest –limb casting –denervation –space flight. Adapted from Faulkner, Green and White In: Physical Activity, Fitness, and Health,Ed. Bouchard, Shephard and Stephens Human Kinetics Publishers, 1994
Endurance training Little hypertrophy but major biochemical adaptations within muscle fibers. Increased numbers of mitochondria; concentration and activities of oxidative enzymes (e.g. succinate dehydrogenase, see below). Succinate dehy- drogenase (SDH) activity: Low activity light High activity dark Control 12-weeks treadmill running Images courtesy of John Faulkner and Timothy White
Calcineurin is a Ca2+-regulated • serine/threonine phosphatase. • Caclineurin dephosphorylates nuclear factor of activated T cells (NFAT) transcription factors. • Dephosphorylated NFATs • translocate to the nucleus where • combinatorially with other factors they activate transcription. • A second target of calcineurin is the transcriptional co-activator, peroxisome-proliferator-activated receptor-g co-activator-1 (PGC-1a). • Activation of calcineurin in skeletal myocytes selectively up-regulates slow-fiber-specific gene promoters and the effect enhanced with PGC-1a expression • PGC-1a activates mitochondrial biogenesis. A calcineurin dependent transcriptional pathway appears to control skeletal muscle fiber type. Lin et al. (2002) Nature 418:797-801
A calcineurin dependent transcriptional pathway appears to control skeletal muscle fiber type. • Cyclosporin is widely used clinically to prevent rejection of transplanted tissues; patients develop skeletal muscle myopathy and loss of oxidative capacity. • Cyclosporin (and FK-506) are specific inhibitors of calcineurin • and thereby block T cell activation. • Cyclosporin administration to intact animals promotes slow-to-fast fiber transformation.
Increased use: strength training Early gains in strength appear to be predominantly due to neural factors…optimizing recruitment patterns. Long term gains almost solely the result of hypertrophy i.e. increased size.
The PI(3)K/Akt(PKB)/mTOR pathway is a crucial regulator of skeletal muscle hypertrophy/atrophy. • Application of IGF-I to C2C12 myotube cultures induced both increased width and phosphor-ylation of downstream targets of Akt (p70S6 kinase, p70S6K; PHAS-1/4E-BP1; GSK3) but did NOT activate the calcineurin pathway. • Treatment with rapamycin almost completely prevented increase in width of C2C12 myotubes. • Treatment with cyclosporin or FK506 does not prevent myotube growth in vitro or compensatory hypertrophy in vivo • Recovery of muscle weight after following reloading is blocked by rapamycin but not cyclosporin. Rommel et al. (2001) Nature Cell Biology3, 1009.
Disuse causes atrophy -- USE IT OR LOSE IT! Individual fiber atrophy (loss of myofibrils) with no loss in fibers. Effect more pronounced in Type II fibers. “Completely reversible” (in young healthy individuals). ATPase activity: Type I fibers light Type II fibers dark Control Prolonged bed rest Images courtesy of John Faulkner
Performance Declines with Aging --despite maintenance of physical activity 100 80 60 Performance (% of peak) 40 Shotput/Discus Marathon 20 Basketball (rebounds/game) 0 20 30 40 50 60 10 Age (years) D.H. Moore (1975) Nature 253:264-265. NBA Register, 1992-1993 Edition
Number of motor units declines during aging - extensor digitorum brevis muscle of human beings AGE-ASSOCIATED ATROPHY DUE TO BOTH… Individual fiber atrophy (which may be at least partially preventable and reversible through exercise). Loss of fibers (which as yet appears irreversible). Campbell et al., (1973) J Neurol Neurosurg Psych 36:74-182.
Motor unit remodeling with aging Central nervous system Muscle Motor neuron loss AGING • Fewer motor units • More fibers/motor unit
Mean Motor Unit Forces: • FF motor units get smaller in old age and decrease in number • S motor units get bigger with no change in number • Decreased rate of force generation and POWER!! 225 200 Adult 175 Old 150 125 Maximum Isometric Force (mN) 100 75 50 25 0 FF FI FR S Kadhiresan et al., (1996) J Physiol 493:543-552. Motor Unit Classification
Muscle injury may play a role in the development of atrophy with aging. • Muscles in old animals are more susceptible to contraction- • induced injury than those in young or adult animals. • Muscles in old animals show delayed and impaired recovery • following contraction-induced injury. • Following severe injury, muscles in old animals display • prolonged, possibly irreversible, structural and functional • deficits.
Only lengthening contractions result in damaged fibers * control 20 passive isometric lengthening * different from zero (p<0.05) 15 Injured fibers (% total) 10 5 0 Other Measures of Contraction-Induced Injury • immediate mechanical disruption observed by EM. • enzyme release from degenerating muscle fibers • in human beings, subjective reports of muscle soreness • in the absence of fatigue, a decrease in the development of force Koh & Brooks (2001) Am J Physiol 281:R155-R161.
“Ghost” fiber 3 days after initial injury Faulkner, Brooks and Zerba (1995) J Gerontol 50:B124-B129.
Perry and Rudnicki (2000) Frontiers in Bioscience 5:D750-67. 4 days after damage 2 weeks after damage 4 weeks after damage with irradiation Repair through activation of satellite cells Myology (Sanes, McGraw-Hill, 1994)
2 weeks post * p < 0.05 * * A single prior exposure to a protocol of lengthening contractions reduced the force deficit and damaged fibers 60% non-trained 50 40 30 20 10 0 Force Deficit (% control) Injured Fibers (% total) Koh & Brooks (2001) Am J Physiol 281:R155-R161.
60 60 50 50 40 40 30 30 20 20 10 10 0 0 Degeneration-regeneration not necessary to provide muscles protection from contraction-induced injury non-trained trained passive trained isometric *different from non- -trained (p<0.05) * * Force deficit (% control) Injured fibers (% total) * Koh & Brooks (2001) Am J Physiol 281:R155-R161. Force deficit Injured fibers • Despite the increase in susceptibility to injury with aging, • and the decreased ability to recover, muscles in old • animals can be conditioned for protection from injury. • Maintenance of conditioned fibers, particularly in muscles • of elderly people, may prevent inadvertent damage during • contractions.
Microstructure Modified from Squire, Muscle: Design, Diversity, and Disease Benjamin/Cummings, 1986 Originally from Lazarides (1980) Nature 283:249-256.
Muscular Dystrophy: A frequently fatal disease of muscle deterioration • Muscular dystrophies have in the past been classified based on subjective and sometimes • subtle differences in clinical presentation, such as age of onset, involvement of particular • muscles, rate of progression of pathology, mode of inheritance. • Since the discovery of dystrophin, numerous genetic disease loci have been linked to protein • products and to cellular phenotypes, generating models for studying the pathogenesis of the • dystrophies. • Proteins localized in the nucleus, cytosol, cytoskeleton, sarcolemma, and ECM. Cohn and Campbell (2000) Muscle Nerve 23:1459-1471.
Dystrophin function: transmission of force to extracellular matrix DGC dystrophin dystroglycan (a and b) sarcoglycans (a, b, g, d) syntrophins (a, b1) dystrobrevins (a, b) sarcospan laminin-a2 (merosin) (Some components of the dystrophin glycoprotein complex are relatively recent discoveries, so one cannot assume that all players are yet known.) Cohn and Campbell (2000) Muscle Nerve 23:1459-1471.