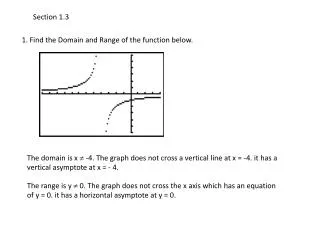
Section 1.3
Section 1.3. The Process of a Statistical Study. Objectives. Describe the process of a statistical study. Identify various types of studies . . Statistical Study . Conducting a Statistical Study 1. Determine the design of the study. a. State the question to be studied.

Section 1.3
E N D
Presentation Transcript
Section 1.3 The Process of a Statistical Study
Objectives Describe the process of a statistical study. Identify various types of studies.
Statistical Study Conducting a Statistical Study 1. Determine the design of the study. a. State the question to be studied. b. Determine the population and variables. c. Determine the sampling method. 2. Collect the data. 3. Organize the data. 4. Analyze the data to answer the question.
Example 1.11: Identifying Population and Variables Neurologists want to study the effect of vitamin C on nerve disorders. The goal of the study is to see if taking an intravenous dose of vitamin C will reduce the amount of nerve pain reported by patients. Identify the population of interest and the variables in this study.
Example 1.11: Identifying Population and Variables (cont.) Solution Since the study seeks to determine if a new treatment will reduce nerve pain in patients with nerve disorders, the population of the study would be limited to patients with these specific types of disorders. Most likely, the study will further narrow the population to focus only on patients with a specific nerve disorder and not group all such patients in the same category. The variables of interest are the amount of vitamin C administered to a patient and the amount of nerve pain each patient reports.
Statistical Study An observational study observes data that already exist. An experiment generates data to help identify cause-and-effect relationships.
Example 1.12: Identifying Observational Studies and Experiments Which type of study would you conduct: an observational study or an experiment? a. You want to determine the average age of college students across the nation. b. Researchers wish to determine if flu shots actually help prevent severe cases of the flu.
Example 1.12: Identifying Observational Studies and Experiments (cont.) Solution a. An observational study would be used since you just need to consider existing records of college students to determine the average age of college students. b. An experiment would need to be used in order to establish a cause-and-effect relationship between flu shots and flu prevention.
Observational Studies Representative Sample A representative sample has the same relevant characteristics as the population and does not favor one group from the population over another.
Example 1.13: Identifying Sampling Methods Identify the type of sampling used in each of the following scenarios. a. A pollster surveys 50 people in each of a senator’s 12 voting precincts. b. The quality control department at a cereal manufacturer measures the weight of every 10th box off of the assembly line. c. A female student walks down the halls in her dorm asking students how much money they would spend in a food court in the dorm lobby in an effort to persuade the administration to offer such an option.
Example 1.13: Identifying Sampling Methods (cont.) d. An educator chooses 5 of the school districts in the Chicago area and asks each household in those districts how many school-age children are in the home. e. To determine who will win a $100,000 shopping spree at the mall, the manager draws a name out of a box of entries.
Example 1.13: Identifying Sampling Methods (cont.) Solution a. Stratified sampling: The voting precincts are the strata. b. Systematic sampling: The system of selecting the sample is to choose every 10th box. c. Convenience sampling: This would be a very easy method of surveying for this particular scenario, and it would provide a representative sample of the dorm residents.
Example 1.13: Identifying Sampling Methods (cont.) d. Cluster sampling: Every school district in the Chicago area is a cluster. e. Random sampling: Every name in the box has a equal chance of being chosen.
Example 1.14: Classifying Studies as Cross-Sectional or Longitudinal Categorize the following studies as either cross-sectional or longitudinal. a. A group of 220 patients is followed for 15 years in order to determine the long-term health effects resulting from gastric bypass surgery. b. A gastroenterologist surveys 130 of his patients six months after having gastric bypass surgery to determine the average amount of weight lost.
Example 1.14: Classifying Studies as Cross-Sectional or Longitudinal (cont.) Solution a. For this study, a group of gastric bypass patients is followed for a period of time. By definition, this is a longitudinal study. b. In this study, a snapshot of the amount of weight lost at a specific point in time is gathered; thus, this is a cross-sectional study.
Types of Observational Studies In a cross-sectional study, data are collected at a single point in time. In a longitudinal study, data are gathered by following a particular group over a period of time. A meta-analysis is a study that compiles information from previous studies. A case study looks at multiple variables that affect a single event.
Example 1.15: Classifying Studies as Meta-Analysis or Case Study Categorize the following studies as either a meta‑analysis or a case study. a. Oceanographers study research on tsunamis dating from 1900 to 2000 to determine their effects on the ocean floor. b. Meteorologists study the Indian Ocean tsunami of December 2004 to try to identify warning signs.
Example 1.15: Classifying Studies as Meta-Analysis or Case Study (cont.) Solution a. Because the oceanographers are looking at multiple studies relating to the single variable of tsunamis’ effects on the ocean floor, this is a meta-analysis study. b. In order to identify tsunami warning signs, meteorologists would most likely look at multiple variables relating to the 2004 tsunami. Because they are studying several aspects of a single tsunami, it is a case study.
Experiments A treatment is some condition that is applied to a group of subjects in an experiment. Subjects are people or things being studied in an experiment. Participants are people being studied in an experiment. The response variable is the variable in an experiment that responds to the treatment. The explanatory variable is the variable in an experiment that causes the change in the response variable.
Experiments Principles of Experimental Design 1. Randomize the control and treatment groups. 2. Control for outside effects on the response variable. 3. Replicate the experiment a significant number of times to see meaningful patterns.
Experiments A control group is a group of subjects to which no treatment is applied in an experiment. A treatment group is a group of subjects to which researchers apply a treatment in an experiment. Confounding variables are factors other than the treatment that cause an effect on the subjects of an experiment.
Experiments The placebo effect is a response to the power of suggestion, rather than the treatment itself, by participants of an experiment. A placebo is a substance that appears identical to the actual treatment but contains no intrinsic beneficial elements.
Experiments In a single-blind experiment, subjects do not know if they are in the control group or the treatment group, but the people interacting with the subjects in the experiment know in which group each subject has been placed. In a double-blind experiment, neither the subjects nor the people interacting with the subjects know to which group each subject belongs.
Example 1.16: Analyzing an Experiment Consider the study from Example 1.11, in which neurologists want to determine if taking an intravenous dose of vitamin C will reduce the amount of nerve pain reported by patients. Suppose that the study was narrowed to focus only on patients with the nerve disorder, multiple sclerosis (MS). After study approval, the neurologists solicit volunteers who are patients with MS who are reporting nerve pain. The participants are then randomly assigned to two groups, each having 20 participants.
Example 1.16: Analyzing an Experiment (cont.) Participants in Group A are administered intravenous doses of vitamin C, and their nerve pain is tracked. Participants in Group B are administered intravenous doses of saline (which has no active ingredients) and their pain levels are also tracked. The patients are not told which of the two groups they are in; however, the nurses administering the IVs are aware of the group assignments. After a predetermined length of time, the amounts of pain reported by the separate groups are compared to determine if an intravenous dose of vitamin C will reduce the amount of nerve pain.
Example 1.16: Analyzing an Experiment (cont.) a. Identify the explanatory and response variables. b. What is the treatment? c. Which group is the treatment group and which group is the control group? d. What is the purpose of administering saline to Group B? e. Is this a single-blind or double-blind study? Do you think this is the best choice for this study?
Example 1.16: Analyzing an Experiment (cont.) Solution a. The explanatory variable is what “explains” the changes in the response variable. Since the neurologists are trying to determine if the dose of vitamin C can reduce nerve pain, the explanatory variable is the dose of vitamin C and the response variable is the amount of nerve pain reported by each patient. b. The treatment is what is being applied to the group, so the treatment is the dose of vitamin C.
Example 1.16: Analyzing an Experiment (cont.) c. The group that received the treatment of vitamin C, namely Group A, is the treatment group. The group that did not receive the treatment, Group B, is the control group. d. The saline that is administered to Group B is a placebo, and is administered to compensate for the placebo effect, so that all patients are responding to the same suggestion that they are receiving treatment.
Example 1.16: Analyzing an Experiment (cont.) e. Since the patients do not know the group assignments, but the nurses who are interacting with the patients do know to which group the patients were assigned, this is a single-blind study. As reported pain is a subjective measure, it would be easy for nurses to unintentionally influence the patients’ responses; thus a double-blind study would probably be a better design for this experiment.
Institutional Review Boards An Institutional Review Board (IRB) is a group of people who review the design of a study to make sure that it is appropriate and that no unnecessary harm will come to the subjects involved. Informed consent involves completely disclosing to participants the goals and procedures involved in a study and obtaining their agreement to participate.