Zinc-Zn
Zinc-Zn. Brenda 7.4. General Info. Atomic number: 30 Melting Point: 419.58 C Boiling Point: 907 C Element Classification: Metal. Discovery. First produced in the 1400s in India Was rediscovered by Andreas Sigismund Marggraf in 1764. Description. It is a soft metal


Zinc-Zn
E N D
Presentation Transcript
Zinc-Zn Brenda 7.4
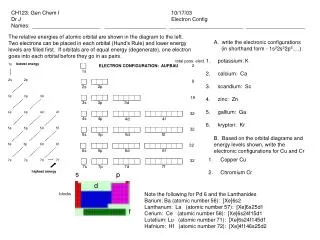
General Info • Atomic number: 30 • Melting Point: 419.58 C • Boiling Point: 907 C • Element Classification: Metal
Discovery • First produced in the 1400s in India • Was rediscovered by Andreas Sigismund Marggraf in 1764
Description • It is a soft metal • It a has grayish or blue-white color • It contains 21 isotopes where 5 of them is stable and the other 16 are not (unstable) • It is brittle at low temperatures but malleable at 100-150 C • It is an electric conductor • It burns in air at high red heat
1. Form numerous alloys Such as… • Brass • Bronze • Nickel Silver • Soft Solder • German Silver • Spring Brass • Aluminum Solder
2. Make die castings In.. • Electrical • Automotive • Hardware
3. To galvanize other metals • To prevent corrosion • Galvanize means to put coating on iron • To make metals stop rusting for quite a long time
Interesting Facts Zinc is.. • Found on roofs because it has a very high quality of roofing material • A metal we all need for life • Part of enzymes and many biological medicated processes • Easily reacted with acids to produce hydrogen • Very slow to oxidize • An interesting element because it is not a poisonous element, where the two other elements that are in the same group with Zinc, Mercury and Cadmium, are both pretty poisonous. • Something we need for us to survive
Hazardous Effects of Zinc to the Environment Zinc production can cause: • Water pollution-it can cause for fish to be accumulated with Zinc which is able to bio magnify up the food chain • Soil pollution-farmlands could be polluted which can cause farm animals to absorb concentration that damages their health. It could also break down microorganisms and earthworms since their habitats are polluted. • Plants to have their system unstable, unhandled and uncontrolled
More information about Zinc • In our body, if we don’t have enough Zinc, we are not able to smell things. • When you combine it with other elements, it could get quite frisky • Zinc is an abundant metal • An enzyme called ‘Carbonic Anhydrase’ needs Zinc to catalyze the reaction of CO2 and water
Bibliography Websites • Helmenstine, Anne Marie. “Zinc Facts.” About.com. 25 April 2010 http://chemistry.about.com/od/elementfacts/a/zinc.htm • “Zinc-Zn.” Lenntech. 25 April 2010 http://www.lenntech.com/periodic/elements/zn.htm • “The Element Zinc.” Jefferson Lab. 25 April 2010 http://education.jlab.org/itselemental/ele030.html Images • “Andreas Sigismund Marggraf.” Photo. 25 April 2010 http://www.bbaw.de/akademie/kalender/biog-pic-009-marggraf.jpg • “Zinc.” Photo. 25 April 2010 http://z.about.com/d/chemistry/1/0/w/c/zinc.jpg • “Zinc Crystal.” Photo. 25 April 2010 http://www.albany.edu/offcourse/july98/zinc_sm.jpg • “Zinc.” Photo. 25 April 2010 http://www.webelements.com/_media/elements/element-pics-theo/30_Zn_1.jpg
“Zinc Rangehood.” Photo. 25 April 2010 http://christopherleeplummer.com/company/blog/wp-content/uploads/2007/09/zinc-rangehood.jpg • “5" Seat Springs (Brass).” Photo. 25 April 2010 http://www.justifieddefiance.com/catalog/images/Brass4Springs.jpg • “Zinc Die Casting-01.” Photo. 25 April 2010 http://www.bldiecasting.com/products_img/200661692428.jpg • “Results for rust.” Photo. 25 April 2010 http://bostonbiker.org/files/2009/12/rust3.jpg • “Roof tiles texture.” Photo. 25 April 2010 http://mayang.com/textures/Architectural/images/Roofing/roof_tiles_2260555.JPG • “Degradation of earth’s land surfaces-land pollution.” Photo. 25 April 2010 http://www.gradebook.org/pollution4.gif • “Carbonic Anhydrase.” Photo. 25 April 2010 http://andromeda.rutgers.edu/~huskey/images/carbonic_anhydrase_active_site_w.png Website (2010, 25 April) Zinc - Periodic Table of Videos Retrieved from http://www.youtube.com/watch?v=99wPiMb-k0o