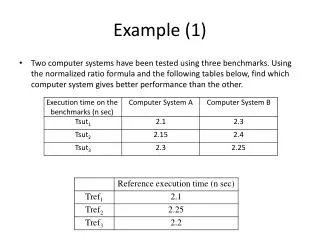
Example 1
Example 1. Sn + HF → SnF 2 + H 2 If you react 62.5 g of hydrofluoric acid with 92.0 g of tin and you produce 109 g of tin (II) fluoride, what was your percent yield?. Answer. Sn + 2 HF → SnF 2 + H 2. 245 g SnF 2. 121 g SnF 2. Finishing.

Example 1
E N D
Presentation Transcript
Example 1 • Sn + HF → SnF2 + H2 • If you react 62.5 g of hydrofluoric acid with 92.0 g of tin and you produce 109 g of tin (II) fluoride, what was your percent yield?
Answer • Sn + 2 HF → SnF2 + H2 245 g SnF2 121 g SnF2
Finishing • Percent yield = actual yield/ theoretical x 100 • 109 g / 121.45… g = • 89.7 %
Example 2 4 4 6 5 • NH3 + O2 NO + H2O • How much NO can be produced from 52 g of NH3 and 79 g of O2 if you have a 84% yield? =92 g NO =59 g NO
Finishing • Don’t round the number until you report your final answer • Actual yield /59.26975 g = .84 • Actual yield = 50. g NO
Question 3 2 2 • HCl + C4H10 C2H6 + Cl2 • You collect 43.5 g of chlorine gas from 84.9 g of HCl and 143 g of C4H10. What was your percent yield? =82.6 g Cl2 =174 g Cl2
Finishing • Actual yield / theoretical yield = percent yield • 43.5 g / 82.5526…g = • 52.7%
Homework • NiS + O2 NiO + SO2 • How many grams of sulfur dioxide gas can be made from 84.9 g of Nickel (II) sulfide and 143 g of oxygen assuming a 68.7% yield?