Water Movement in Plants: Bulk Flow vs. Osmosis
E N D
Presentation Transcript
Osmosis Plant Physiology 2009 UNI
Two ways to move water • Bulk flow • Osmosis • Both move water from high energy to low • Differences • Source of energy difference • What gets moved • Structures
Bulk flow • Source of energy difference: Δpressure • Requires open “pipes” • Everything in the fluid moves at once • Advantages • Move lots of material • Move long distances quickly • Disadvantage • No chance to select what gets moved
Examples of bulk flow Xylem sap is being pulled up. Phloem sap is being pushed down. Heart contraction pushes blood out into vessels.
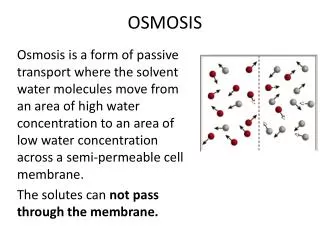
Osmosis • Source of energy difference: • Solutes control energy (potential) of water • Pressure also controls energy (potential) of water • Requires selective membrane between compartments • Lets water through • Does not let solutes through • Examples: water movement between adjacent cells • Restoration of turgor to wilted leaves • Rehydration of cells of marathon runner • Advantage • Lets organism move (just) the water • Disadvantages • Slow • Small volumes moved • Short distances
Lowering water energy with solutes • Start with pure water • Maximum water energy (potential) • Effect of solutes on water energy • Solutes partly tie up water • Solutes reduce water energy • Can water energy by adding solutes • Solute effect on water energy • 0 if no solutes • Negative if solutes present • More solutes? More negative
Seawater pure water Example I of simple osmosis • Water permeable bag allows water to move but not salts • Water moved • From high energy (pure water) • To low energy • Continues until no water left in bag • No pressure differences between bag and surroundings air Seawater air pure water START LATER
fresh water salty water Example II of simple osmosis • Water permeable bag allows water to move but not salts • Water moved • From high water energy (pure water) • To low water energy (water tied up by solutes) • Continues until bag breaks • No pressure differences between bag and surroundings air fresh water air salty water START LATER
Osmotic movement of water • Continues until • energy of water = in both compartments • then molality (not molarity) will be the same in both • or a pressure difference develops • doesn’t happen with flexible bag, animal cells • does happen with stiff cell walls (coming up) • No energy difference = no net movement
Changing water energy with pressure • Start with pure water • Maximum water energy (potential) • Effect of pressure • Can by increasing pressure • Can by lowering pressure • Pressure can be +, 0, - • Pressure-induced differences add to solute-induced differences
Example I of cell osmosis Seawater air Seawater air • Water permeable bag allows water to move but not salts • Water moves • From high energy (pure water) to low energy • Until concentration of solutes (water energy) = inside and out • No pressure differences between bag and surroundings • Cell with membrane shrinks away from wall=plasmolysis typical cell typical cell LATER cell wall START
Example II of cell osmosis • Water permeable bag allows water to move but not salts • Water moved • From high energy (pure water) to low energy • Continues until pressure effect on water energy = solute effect • Big pressure differences between bag and surroundings • Cell volume hardly changes (pressure does) fresh water air fresh water air typical cell typical cell START LATER