X-rays
Physics 102: Lecture 26. X-rays. Make sure your grade book entries are correct. But first a quick review of the periodic table. http://www.youtube.com/watch?v=SmwlzwGMMwc. http://www.youtube.com/watch?v=GFIvXVMbII0&feature=related. X-Rays.

X-rays
E N D
Presentation Transcript
Physics 102: Lecture 26 X-rays Make sure your grade book entries are correct.
But first a quick review of the periodic table http://www.youtube.com/watch?v=SmwlzwGMMwc http://www.youtube.com/watch?v=GFIvXVMbII0&feature=related
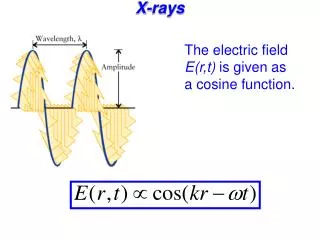
X-Rays Photons with energy in approx range 100eV to 100,000eV. This large energy means they go right through you (except for your bones). What are the wavelengths? .01 nm to 10 nm
X-Ray Production How do you produce 100 eV photons? • Black Body Radiation • Would require temperature over 10 times hotter than surface of sun • Excitation of outer electrons • Typically have energy around 10 eV • Radioactive Decays • Hard to turn on/off
Recall from Lecture 3: EPE = V q KE = EPE = (70,000 V) (1 e-) = 70,000 eV = 11.2 x 10-14 J = 1.6 x 10-19 C Electron Tubes • Accelerate an electron through a voltage difference to give it some energy... Example An electron is accelerated through a potential difference of 70,000 V. How much energy does it emerge with? EPE of voltage gap becomes K.E. for electron.
From Electrons to X-Rays • Now take these high energy electrons (up to 100,000 eV) and slam them into heavy atoms - any element. • 2 kinds of X-Rays are produced: • “Bremsstrahlung” • “Characteristic” VIS
intensity 0 Bremsstrahlung X-Rays • Electron hits atom and slows down, losing kinetic energy. • Energy emitted as photon • Electron hitting atom makes many photons (X-Rays), all with different energy. • Many different wavelengths. • If all of electron’s energy is lost to a single photon, photon has maximum energy (minimum wavelength). • Minimum X-Ray wavelength = lo.
An electron is accelerated through 50,000 volts What is the minimum wavelength photon it can produce when striking a target? Minimum wavelengthMaximum energy intensity 0 Example Bremsstrahlung Practice Electron loses ALL of its energy in one collision and emits one photon.
L shell (n=2) e- e- e- K shell (n=1) e- e- e- (high energy electron) Characteristic X-Rays Electron knocks one of the two K shell (ground state) electrons out of an atom. L (n=2) or higher shell electron falls down to K shell (ground state) and x-ray photon is emitted Characteristic x-ray nomenclature n=1 “K shell” n=2 “L shell” n=3 “M shell”
e- ejected electron e- Characteristic X-Rays e- Electron knocks one of the two K shell (ground state) electrons out of an atom. L (n=2) or higher shell electron falls down to K shell (ground state) and x-ray photon is emitted L shell (n=2) e- e- e- Characteristic x-ray nomenclature n=1 “K shell” n=2 “L shell” n=3 “M shell” K shell (n=1) e- e-
e- L shell electron falls down Characteristic X-Rays e- X-Ray photon emitted “K X-ray” (n=2 n=1 transition) Electron knocks one of the two K shell (ground state) electrons out of an atom. L (n=2) or higher shell electron falls down to K shell (ground state) and x-ray photon is emitted L shell (n=2) e- e- e- Characteristic x-ray nomenclature n=1 “K shell” n=2 “L shell” n=3 “M shell” K shell (n=1) e- e-
Better formula for multi-electron atoms Careful! the formula n=2 assumed a single electron bound to just a positive nucleus. L n=1 Ka photon K intensity (vs. Expt) Example Ka X-Rays Estimate the energy of Ka X-rays off of a silver (Ag) target (Z=47). Not bad!
Ka X-rays come from n=2 n=1 transition. What about n=3 n=1 transition? Kb X-Rays are higher energy (lower l) than Ka.(and lower intensity) Ka Kb intensity Kb X-Rays Not as likely, but possible. Produces Kb X-Rays! Different elements have different Characteristic X-Rays
Ka Kb intensity 0 Ka Kb intensity All Together Now... Brehmsstrahlung X-Rays and Characteristic X-Rays both occur at the same time. intensity 0
K K Kb Kb intensity intensity Preflight 26.1 These two plots correspond to X-Ray tubes that: (1) Are operating at different voltages (2) Contain different elements (3) Both (4) Neither
intensity intensity ACT: X-Rays I Which graph corresponds to the tube being operated at the higher voltage? 1) Top 2) Bottom
intensity intensity ACT: X-Rays II The top spectrum comes from a tube with a silver target (Ag, Z=47). What is the bottom target? 1) Pd, Z=46 2) Ag, Z=47 3) Cd, Z=48
From atoms to nuclei to nucleons to quarks: The heirarchy of sizes
Nuclear Physics A Z Nucleus = Protons+ Neutrons nucleons Z = proton number (atomic number) Gives chemical properties (and name) N = neutron number A = nucleon number (atomic mass number) Gives you mass density of element A=N+Z Periodic_Table
Preflight 27.1 A material is known to be an isotope of lead Based on this information which of the following can you specify? 1) The atomic mass number 2) The neutron number 3) The number of protons
Strong Nuclear Force • Rutherford experiment shows that all the positive charge is contained in a small nucleus • Size ~ few x 10-15 m (few fm) • Let’s estimate EPE of two protons separated by 1 fm EPE = kq2/r = (9 x 109)(1.6 x 10-19)2/10-15 = 2.3 x 10-13 J = 1.44 x 106 eV = 1.44 MeV • Therefore, the force that binds protons and neutrons together to form a nucleus must be very strong in order to overcome Coulomb repulsion • But the force acts over very short distances—of order few fm Two atoms don’t feel force
Coulomb force proton electron proton neutron Very strong force Binding energy ofdeuteron=or 2.2Mev!That’s around 200,000 times bigger! Strong Nuclear Force Hydrogen atom:Binding energy=13.6eV (of electron to nucleus) Simplest Nucleus: Deuteron=neutron+proton
Can get 4 nucleons into n=1 state. Energy will favor N=Z # protons = # neutrons Pauli Principle - neutrons and protons have spin like electron, and thus ms= 1/2. But protons repel one another (Coulomb Force) and when Z is large it becomes harder to put more protons into a nucleus without adding even more neutrons to provide more of the Strong Force. For this reason, in heavier nuclei N>Z. 7
Nuclei have energy level—just like atoms 12C energy levels 60Ni energy levels Note the energy scale is MeV rather than eV