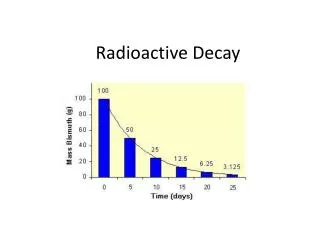
Radio Active decay
This educational outline delves into the fascinating world of radioactive decay, detailing the four primary types of emissions: alpha, beta, positron, and gamma. It explains how nuclear particles interact and transform through these emissions, such as how alpha particles are released when protons and neutrons are emitted, or how beta emissions occur when a neutron converts into an electron. The document concludes with insights on electron capture and gamma emissions that transition nuclei to lower energy states, providing a comprehensive overview of the topic.

Radio Active decay
E N D
Presentation Transcript
Radio Active decay By Stephen Goose 422-004 1206455 Argon
Outline • 1. Title • 2. Outline • 3. Alpha Emissions • 4.Beta Emissions • 5. Positrons Emissions • 6. Electron Capture • 7. Gamma Emissions • 8. Conclusions • 9. Bibliography
Alpha Emissions • When two protons and neutrons are emitted from the nucleus! During radioactive decay
. Beta Emissions • A larger neutron. Proton ratio means it needs to be converted into electrons which are from the nucleus • 0n+1p=-1B
, Positron Emissions • When ratio is stable it is emitted with a mass of a electron but with a positive charge. • 38=38+0 • 19K=18Ar+1B
, Electron Capture • When a electron’s inner orbital is captured its own atom’s nucleus. • 106+0=106 • 47 Ag+-1e=46Pd
, Gamma emissions • When gamma rays are emitted from the nucleus to a ground energy state • No equation
Conclusion • So now that you now everything.. Oh wait you don’t! • Radioactive decay- When a nucleus is made lighter by taking out particles. They all come from the atom.
Bibliography • http://library.thinkquest.org/3471/alpha_decay.gif • http://hyperphysics.phy-astr.gsu.edu/hbase/nuclear/beta.html • http://education.jlab.org/glossary/betadecay.gif • http://search.msn.com/images/results.aspx?q=electron+capture&FORM=MSNH&mkt=en- • US#focal=0e0dc41af3c974b2444bf591fb1a6859&furl=http%3A%2F • http://search.msn.com/images/results.aspx?q=gamma+emission+ • &go=Search+Images&form=QBIR#focal=b96cc2acdb02dcac638122fe1ff97f7d&furl=http%3A%2 • F%2Fwww.euronuclear.org%2Finfo%2Fencyclopedia%2Fimages%2Fang_zue.jpg • http://www.dallassd.com/our%20schools/high%20School/Chemsite/nucleareqns/ttypes3.html • %2Fwww.euronuclear.org%2Fimages%2Felektroneneinfange.jpg • US#focal=0e0dc41af3c974b2444bf591fb1a6859&furl=http%3A%2F • http://education.jlab.org/glossary/betadecay.gif • http://library.thinkquest.org/3471/alpha_decay.gif • http://www.dallassd.com/our%20schools/high%20School/Chemsite/nucleareqns/ttypes3.html • http://search.msn.com/images/results.aspx?q=electron+capture&FORM=MSNH&mkt=en- • US#focal=0e0dc41af3c974b2444bf591fb1a6859&furl=http%3A%2F