RADIOACTIVE DECAY
RADIOACTIVE DECAY. WHAT MAKES AN ELEMENT RADIOACTIVE?. It has an unstable atomic nucleus. A nucleus is only stable with the right number of protons and neutrons An unstable nucleus will undergo radioactive decay giving off invisible waves of energy in the process.

RADIOACTIVE DECAY
E N D
Presentation Transcript
WHAT MAKES AN ELEMENT RADIOACTIVE? • It has an unstable atomic nucleus. • A nucleus is only stable with the right number of protons and neutrons • An unstable nucleus will undergo radioactive decay giving off invisible waves of energy in the process.
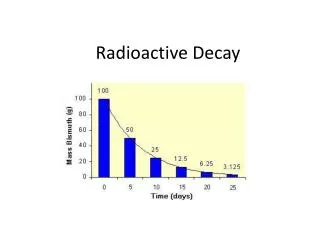
The original radioactive element is called the parent. • The nonradioactive element that forms as the parent element decays is a stable element called the daughter. • The amount of time it takes for half the parent element to turn into the daughter element is called a half-life.
An isotope is a form of an element that differs in numbers of neutrons.
CARBON 14 (RADIOCARBON) • radioactive form of carbon • present in all living things • after the organism dies, the carbon 14 is not renewed and decays at a uniform rate
The half-life of carbon 14 is 5,730 years. After that time, half of the atoms of carbon 14 have turned into nitrogen 14. In another 5,730 years, half the remaining carbon 14 decays (1/4 of the original carbon 14 is left.) • Anything older than about 50,000 years cannot be accurately dating using carbon 14.