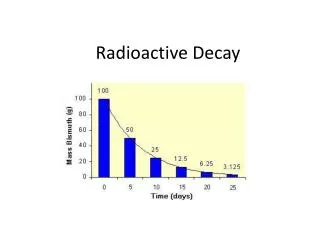
Radioactive Decay
Radioactive Decay. What happens to the nucleus of an atom when it emits a radiation?. Starter : Think about the reaction between Mg and O BRIGHT WHITE LIGHT What is happening? Too easy? Think about the particles. . MgO. Magnesium loses 2 electrons – gives them to oxygen

Radioactive Decay
E N D
Presentation Transcript
Radioactive Decay What happens to the nucleus of an atom when it emits a radiation? Starter: Think about the reaction between Mg and O BRIGHT WHITE LIGHT What is happening? Too easy? Think about the particles.
MgO Magnesium loses 2 electrons – gives them to oxygen Oxygen therefore gains 2 electrons. ~ a simple example showing that atoms can gain / lose electrons in NORMAL Reactions.
Learning Outcomes • H: complete equations to show the effect on the nucleus of the emission of alpha or beta particles • H: explain what isotopes are.
Isotopes • Isotopes are atoms of the same element with different mass numbers • That is; • Same Atomic Number • Same no. of protons • Different Mass Number • Different no. of neutrons
Isotopes of hydrogen Tritium Protium Deuterium 1 proton, 0 neutron, 1 electron 1 proton, 1 neutron, 1 electron 1 proton, 2 neutron, 1 electron
Alpha Decay • When an unstable nucleus emits an alpha particle it loses 2 protons and 2 neutrons For example, Radium 226 decays by alpha emission 226 222 4 Ra Rn + He 88 86 2 Note: The atomic and mass numbers on both sides of the equation balance.
Beta decay – more complex • A beta particle is an electron. But where does this electron come from? • When an unstable nucleus emits an electron, a neutron in its nucleus changes into a proton and an electron. The electron is emitted. • For example Polonium-218 decays by beta emission 218 218 0 Po At e + 84 85 -1 Note: The atomic and mass numbers on both sides of the equation balance.
Gamma Decay • After an alpha or beta particle has been emitted from the nucleus of an isotope, the nucleus has too much energy. • To get rid of that excess energy, a gamma wave is emitted.
Summary • Alpha Decay • Beta Decay • Gamma Decay a wave of energy 226 222 4 Ra Rn + He 88 86 2 218 218 0 Po At e + 84 85 -1
Task • Complete the card sort “decay series”
Task: • produce a poster showing clearly the types of nuclear decay. • C- explain each type of decay • B – give chemical examples which show the number of p, n and e before and after decay. • A – describe the relative dangers of each type of ionising radiation linking each to what they are made of.
Plenary – Block Busters • http://www.teachers-direct.co.uk/resources/quiz-busters/quiz-busters-game.aspx?game_id=10700