Radioactive Decay
Radioactive Decay. Predictions – Chapter 1, Activity 8. Radioactive Decay. Some configurations of protons and neutrons in the nucleus are not as stable as others. When the nucleus is not stable, it will spontaneously transform into a more stable configuration.

Radioactive Decay
E N D
Presentation Transcript
Radioactive Decay Predictions – Chapter 1, Activity 8
Radioactive Decay • Some configurations of protons and neutrons in the nucleus are not as stable as others. • When the nucleus is not stable, it will spontaneously transform into a more stable configuration. • For example, Carbon-14 is not stable and will therefore spontaneously decay into Nitrogen-14. • The process by which an atom spontaneously changes into a more stable one is called radioactive decay.
Unstable 14C14N + e- 6 7 Stable 12C 6 Radio-Isotopes • Isotopes are configurations of atoms that have the same number of protons, but different numbers of neutrons. • Radio-isotopes are unstable isotopes that go through radio active decay.
Types of Radioactive Decay • Alpha Decay • Beta Minus Decay • Beta Plus (Positron) Decay • Gamma Decay
Parent Atom Daughter Atom Alpha Decay Alpha Decay occurs when two protons and two neutrons (or a helium nucleus) are emitted from the nucleus of the parent atom.
Parent Atom Daughter Atom Beta Minus Decay Beta Minus Decay occurs when a neutron decays into a proton and an electron, where the latter is emitted from the nucleus.
Parent Atom Daughter Atom Beta Plus Decay Beta Plus Decay occurs when a proton decays into a neutron and a positive electron (positron), where the latter is emitted from the nucleus.
Alpha particles can usually be stopped by a very thin barrier. Radioisotopes emitting alpha particles are usually not hazardous outside the body, but they can cause damage if ingested. Betas (streams of electrons) can pass through a hand, but are usually stopped by a modest barrier such as a few millimeters of aluminum, or even a layer of clothing. As with alphas, beta particles are more hazardous if inhaled or ingested. Gammas can be very penetrating and can pass through thick barriers. Several feet of concrete would be needed to stop some of the more energetic gammas. A natural gamma source found in the environment (and in the human body) is 40K, an isotope of potassium. Neutrons are also very penetrating. Some elements, like hydrogen, capture and scatter neutrons. Water is commonly used as a neutron radiation shield.
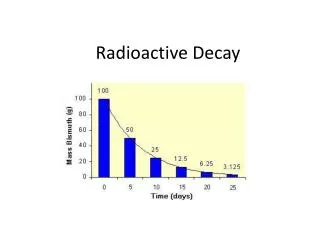
14C14N + e- 6 7 Half-Life • 1 half-life equals the amount of time required for ½ of the original number of parent atoms to decay into daughter atoms.
Example: • You have 600 grams of iodine-133. The half-life of iodine-133 is 21 hours. • How much of this sample would exist after 21 hours? • After 42 hours? • After 126 hours? • Remember: the amount of radioactive substance diminishes by ½ for every half-life.
Example 1: (cont.) • Begin by determining the number of half-lives by dividing the elapsed time by the half-life. • 21 hours/21 hours = 1 half-life • 42 hours/21 hours = 2 half-lives • 126 hours/21 hours = 6 half-lives
Example 1: (cont.) • Since the amount of radioactive material will reduce by one-half with every passing half-life, we can conclude: Divider = 2n where n equals the number of half-lifes.
Example 1: (cont.) • For every half-life, divide the original amount of radioactive material by the divider corresponding to the ½-life in question. • When n = 1, the divider equals 2 or 21. m = 600 g/2 = 300 g • When n = 2, the divider equals 4 or 22. m = 600 g/4 = 150 g • When n = 6, the divider equals 64 or 26. m = 600 g/64 = 9.4 g
Determining the Amount of Radioactive Material Left • To determine the amount of radioactive material left in a sample after n number of ½ - lifes: m = mo 2n Where: m = mass after n ½ - lifes mo = initial mass n = number of ½ - lifes
Applications of Radioactivity • Dating materials: • Carbon-14 (1/2-life = 5730 yrs) used for dating plants and animals that were formerly alive. • Uranium-238 (1/2-life = 4.47 billion years) used for dating rocks. • Potassium-40 (1/2-life = 1.3 billion years) used for dating rocks. • Medical diagnostics. • Cancer Treatment • Smoke detectors.
Carbon Dating • Carbon-14 is created by cosmic radiation from the sun acting on the atmosphere. • CO2 makes up a small portion of our atmosphere that is consumed by plants through photosynthesis. • While all animals and plants are living, Carbon-14 is being constantly replaced. • When a living organism dies, the replenishment of carbon-14 no longer exists and the decay process takes over. • Every 5,730 years, the amount of Carbon-14 will decrease by ½ into Nitrogen-14.
CARBON DATING The rate at which 14C decays is absolutely constant. Given any set of 14C atoms, half of them will decay in 5730 years. Since this rate is slow relative to the movement of carbon through food chains (from plants to animals to bacteria) all carbon in biomass at earth's surface contains atmospheric levels of 14C. However, as soon as any carbon drops out of the cycle of biological processes - for example, through burial in mud or soil - the abundance of 14C begins to decline. After 5730 years only half remains. After another 5730 years only a quarter remains. This process, which continues until no 14C remains, is the basis of carbon dating
Cobalt- 60 Cancer Treatment • Gamma ray emitter • Half life 5.23 years • Radiotherapy – treatment of cancer with radiation • Cancerous cells more susceptible to damage by ionising radiation than normal cells • Ionising radiation is directed on to the tumour from different directions • Tumour dose is high but normal tissue receives a much lower, less harmful dose
Cobalt - 60 • Gamma rays kill micro-organisms in food • May also involve inhibiting sprouting, controlling ripening and pasteurising foods • Fears of genetic modification
Americium-241 • Emitted α-particles ionise the air molecules, conduct an electric current between two terminals • Smoke clings to ionised air molecules and slows them down • Current decreases and a transistor switch activates the alarm • This type of alarm not used these days
NUCLEAR ENERGY FISSION Splitting the Uranium Atom: Uranium is the principle element used in nuclear reactors and in certain types of atomic bombs. The specific isotope used is 235U. When a stray neutron strikes a 235U nucleus, it is at first absorbed into it. This creates 236U. 236U is unstable and this causes the atom to fission
Light atoms tend to combine and release energy as they do so. Heavy atoms tend to split and release energy as they do so. Uranium and Plutonium are particularly useful in this regard, and are the basis of nuclear fission. Note there are three neutrons released for every incident neutron. This is the basis of a chain reaction. http://www.youtube.com/watch?v=JxzPN-vdP_0&feature=related http://www.youtube.com/watch?v=XHitaEy-Xtg
Light atoms tend to combine and release energy as they do so. Heavy atoms tend to split and release energy as they do so. Uranium and Plutonium are particularly useful in this regard, and are the basis of nuclear fission. Heavy nuclei break into lighter nuclei and energy is released. Light nuclei fuse into heavy nuclei and energy is released.
In a nuclear reactor, however, the last thing you (and the rest of the world) want is all your atoms splitting at once. But the reactor core needs to be slightly supercritical so that plant operators can raise and lower the temperature of the reactor. The control rods give the operators a way to absorb free neutrons so operators can maintain the reactor at a critical level.To turn nuclear fission into electrical energy, the first step for nuclear power plant operators is to be able to control the energy given off by the enriched uranium and allow it to heat water into steam