Pulping and Bleaching PSE 476/Chem E 471
290 likes | 650 Vues
Pulping and Bleaching PSE 476/Chem E 471. Lecture #17 Introduction to Bleaching. Agenda. Brightness General Bleaching Principles Chemistry Process Chemicals Description Advantages/Disadvantages. Why Bleach?. Improve brightness. Improve brightness stability.

Pulping and Bleaching PSE 476/Chem E 471
E N D
Presentation Transcript
Pulping and BleachingPSE 476/Chem E 471 Lecture #17 Introduction to Bleaching
Agenda • Brightness • General Bleaching Principles • Chemistry • Process • Chemicals • Description • Advantages/Disadvantages
Why Bleach? • Improve brightness. • Improve brightness stability. • Clean up pulp (impurities). • Wood based (bark, resins, sand, shives). • Process based (carbon specs, rust, rubber). • External sources based (plastics, grease, ash). • Increase capacity of paper to accept printing.
Reflected Light Absorbed Transmitted Brightness Determination (1) Light shinning on a sheet of paper is either transmitted, adsorbed, or reflected. • Light is scattered by fibers at air/fiber interfaces • Light is adsorbed by certain chemicals in the fibers (lignin)
Brightness Determination (2) • Brightness is measurement of how much light is reflected from a sheet of paper. • Whiteness does not mean brightness. • Whiteness is a physical phenomena related to how the eye views the paper. • A very white looking piece of paper may not have high brightness. • Example: blue dye added to a yellow tinged sheet of paper will give a white sheet of paper with low brightness.
Brightness Determination (3) • Brightness determination method: • Light reflectance measured and compared to light reflectance from MgO. • MgO assumed to reflect 100% light. • Brightness is reported as % of MgO reflectance (85 brightness is equivalent to 85% of MgO). • Variables: • Angle of light: Light is applied to sheet at 45° angle. • Wavelength: 457 nm (blue light most sensitive). • Pine kraft • Unbleached-ISO 23-28% • Semi bleached-ISO 60-80% • Bleached-ISO 88-91%
General Principles • Two types of bleaching: • Lignin removing: chemical pulps. • Lignin retaining: mechanical pulps. • Bleaching is used because at a certain point in the pulping process, carbohydrate degradation becoming greater than lignin removal. • Bleaching chemicals are more selective for lignin. • Bleaching chemicals much more expensive than pulping chemicals so they are not used in pulping.
General Principles:Chemistry • Pulping • Pulping typically involves cleavage of ether linkages and some substitution (sulfonation). • Bleaching • Bleaching involves attacks on aromatic rings, olefinic structures, and carbonyl groups. • Substitution reactions play a big role.
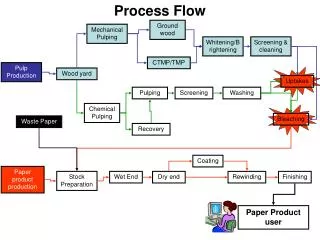
General Principles:Process • Bleaching uses a combination of chemicals in series. • One chemical alone will not remove residual lignin. • Each step reacts with material modified in previous step. NaOH NaOH ClO2 ClO2 O2 O2 Unbleached D D EO EO Bleached Pulp Pulp
Groups of bleaching chemicals (2) • Bleaching chemicals can be divided into three groups according to their function: • 1 Group • The chlorine (Cl2), ozone (O3) reacts with all aromatic lignin units (phenolic groups and their =bonds) • 2 Group • The chlorine dioxide(ClO2) and oxygen (O2) reacts in general with lignin structures that have free phenolic hydroxyl groups • 3 Group • The hypochlorite (H) and hydrogen peroxide (H2O2) reacts only with certain functional groups, for example carbonyl groups
Bleaching reactions • Bleaching chemicals are used primarily as oxidants, to break down residual lignin and to increase its solubility. • Mode of operation: • Electrophiles (oxidative reactions, low pH, involve cations) • Nucelophiles (reductive reactions, high pH, anions) • Radicals
Bleaching Generalities • It is important to note that when bleaching with a specific reagent, it will be converted into a number of different reactive species which will react with lignin and carbohydrates differently. A simple example is when chlorine gas is added to water; both hypochlorous acid and/or hypochorite is formed depending on the pH.