Th pH scale
Th pH scale. What should be written in the three boxes?. Starter – 5 mins. Complete these sentences: Acid rain is… The causes of a cid rain are… The problems with acid rain are… Extension: How can we prevent acid rain?. Acid deposition. Learning outcomes All of you will be able to:


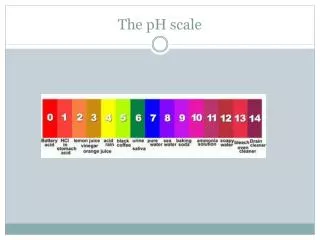
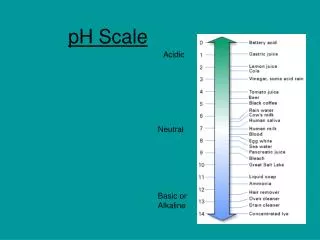
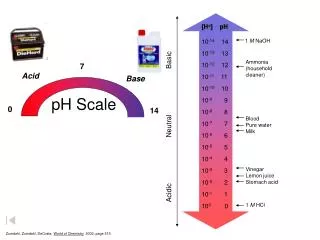
Th pH scale
E N D
Presentation Transcript
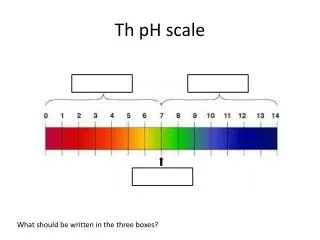
Th pH scale What should be written in the three boxes?
Starter – 5 mins Complete these sentences: • Acid rain is… • The causes of acid rain are… • The problems with acid rain are… Extension: • How can we prevent acid rain?
Acid deposition Learning outcomes All of you will be able to: • Identify the components of acid precipitation Most of you should be able to: • Outline the chemistry leading to the formation of acidified precipitations • Describe 3 possible effects on soil, water and living organisms
Causes Acid deposition forms when we burn fossil fuels. They release: -carbon dioxide -sulphur dioxide -nitrogen oxides
Acid Deposition These gases are released from factories, cars and other vehicles.
The main cause is power stations (which can be used for making electricity), factories and our own homes. • The burning of coals and oil can release sulphur dioxide and nitrogen oxides as gas. Around half of the nitrogen oxide is from cars • These gases react with water vapour in the air, making the rain more acidic • The rain which falls is a weak acid
Acid Deposition Nitrogen oxides and sulphur dioxide cause acid deposition
Sulphur dioxide (SO2) and water (H2O) form sulphurous acid (H2SO3) and sulphuric acid (H2SO4) • Some oxides of nitrogen like nitrogen dioxide (NO2) react with water to form nitrous acid (HNO2) and nitric acid (HNO3) • Carbon dioxide in the air can dissolve in rain water to form carbonic acid, (H2CO3).
Acid Deposition Effects • Direct – weakening tree growth • Toxic effects – effect of aluminium ions on fish and plant roots • Nutrient effects – leaching of nutrients
Nutrients are leached from the soil, pathogens and insects gain entry to the trees. Acid Deposition Acid rain can kill leaves and even the roots, killing the plant Leaves and buds show yellowing (loss of chlorophyll), lesions, thinning of wax cuticle.
Acid precipitation decreases the pH of the soil making aluminium ions more soluble – this aluminium eventually ends up in rivers and lakes Acid Deposition Low concentrations – aluminiumdisturbs the fish’s ability to regulate the amount of salt and water in its body. Higher concentrations – aluminium forms a solid in the fish’s gills leading to suffocation Acid rain can fall into rivers and lakes causing plants and animals to die
Acid deposition can cause limestone and marble to deteriorate (become damaged) • It can also cause damage to the paint on cars if it rains on them for a long time • It causes plants to become damaged and even die, by landing on the leaves as well as affecting the soil • It can affect human health if the water is consumed, or in contact with the skin a lot
StoryBoard – 10 mins Objectives Explainthe process of acid rain using the storyboard in your own words. You must describe how acid rain is madeand the effects it has on the environment.
Clues Electricity is made inpower stationsby steam turning a turbine. When fuels burn they release gases. H2SO4 is the chemical formula for Sulphuric Acid. HNO3 is the chemical formula for Nitric Acid. CaCO3 is the chemical formula for limestone.
Plenary – 5 mins Complete these sentences: • Acid rain is… • The causes of acid rain are… • The problems with acid rain are… Extension: • How can prevent acid rain?