RNA processing #1
RNA processing #1. Making ends of RNA. Types of RNA processing. A) Cutting and trimming to generate ends: rRNA, tRNA and mRNA B) Covalent modification: Add a cap and a polyA tail to mRNA Add a methyl group to 2’-OH of ribose in mRNA and rRNA Extensive changes of bases in tRNA C) Splicing

RNA processing #1
E N D
Presentation Transcript
RNA processing #1 Making ends of RNA
Types of RNA processing • A) Cutting and trimming to generate ends: • rRNA, tRNA and mRNA • B) Covalent modification: • Add a cap and a polyA tail to mRNA • Add a methyl group to 2’-OH of ribose in mRNA and rRNA • Extensive changes of bases in tRNA • C) Splicing • pre-rRNA, pre-mRNA, pre-tRNA by different mechanisms.
Cutting and Trimming RNA • Can use endonucleases to cut at specific sites within a longer precursor RNA • Can use exonucleases to trim back from the new ends to make the mature product • This general process is seen in prokaryotes and eukaryotes for all types of RNA
Excision of mature rRNA and tRNA from pre-rRNA in E. coli tRNA 5S rRNA tRNA Genes: 23S rRNA 16S rRNA Promoters Terminators Transcription 30S pre-rRNA: Cleavage at Further trimming 16S rRNA tRNA 23S rRNA 5S rRNA tRNA
RNase III cuts in stems of stem-loops 16S rRNA 23S rRNA RNase III No apparent primary sequence specificity - perhaps RNase III recognizes a particular stem structure.
Endo- and exonucleases to generate ends of tRNA • Endonuclease RNase P cleaves to generate the 5’ end. • Endonuclease RNase F cleaves 3 nucleotides past the mature 3’ end. • Exonuclease RNase D trims 3’ to 5’, leaving the mature 3’ end.
CCA at 3’ end of tRNAs • Virtually all tRNAs end in the sequence CCA. • Amino acids are added to the CCA end during “charging” of tRNAs for translation. • In most prokaryotic tRNA genes, the CCA is encoded in the DNA. • For most eukaryotic tRNAs, the CCA is added after transcription, in a reaction catalyzed by tRNA nucleotidyl transferase.
Where is the catalytic activity in RNase P? RNase P is composed of a 375 nucleotide RNA and a 20 kDa protein. The protein component will NOT catalyze cleavage on its own. The RNA WILL catalyze cleavage by itself !!!! The protein component aids in the reaction but is not required for catalysis. Thus RNA can be an enzyme. Enzymes composed of RNA are called ribozymes.
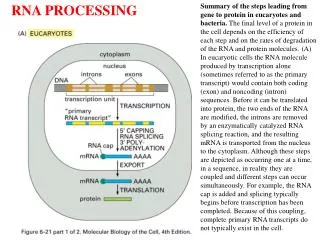
5’ and 3’ ends of eukaryotic mRNA Add a GMP. Methylate it and 1st few nucleotides Cut the pre-mRNA and add A’s
3' 5' OH g G P 5' G b b a P P P-P-P P OH a a b P P 5' to 5' link P GTP A A a P P P A C C P P P C U U P P P g U G G P PPi P RNA being RNA being G synthesized synthesized RNA being synthesized OH OH 3' 3' mRNA guanylyl RNA OH 3' transferase triphosphatase (P=phosphoryl) methyl transferases Add methyl groups to N7 of capping G, 2' OH of 1st and sometimes 2nd nucleotides, etc. Synthesis of 5’ cap
Cleavage and polyadenylation at the 3’ end Cut site CPSF = Cleavage and polyadenylation specificity factor CstF = cleavage stimulation factor CFI, CFII = cleavage factors PAP = polyA polymerase
RNA is processed while being synthesized • Tight linkage between transcription and processing • Processing proteins associated with CTD of large subunit of RNA polymerase II: • Capping enzymes: mRNA guanylyl transferase, methyl transferases • Cleavage and polyadenylation factors: CPSF, CstF • Splicing factors: SR proteins to recruit spliceosomes (next class) • Can visualize splicing on nascent transcripts in EM
Functions of 5’ cap and 3’ polyA • Both cap and polyA contribute to stability of mRNA: • Most mRNAs without a cap or polyA are degraded rapidly. • Shortening of the polyA tail and decapping are part of one pathway for RNA degradation in yeast. • Need 5’ cap for efficient translation: • Eukaryotic translation initiation factor 4 (eIF4) recognizes and binds to the cap as part of initiation.
Splicing of RNA Overview of types of splicing
4 major types of introns • 4 classes of introns can be distinguished on the basis of their mechanism of splicing and/or characterisitic sequences: • Introns in pre-tRNA • Group I introns in fungal mitochondria, plastids, and in pre-rRNA in Tetrahymena • Group II introns in fungal mitochondria and plastids • Introns in pre-mRNA
Splicing of pre-tRNA • Introns in pre-tRNA are very short (about 10-20 nucleotides) • Have no consensus sequences • Are removed by a series of enzymatic steps: • Cleavage by an endonuclease • Phosphodiesterase to open a cyclic intermediate and provide a 3’OH • Activation of one end by a kinase (with ATP hydrolysis) • Ligation of the ends (with ATP hydrolysis) • Phosphatase to remove the extra phosphate on the 2’OH (remaining after phosphodiesterase )
Steps in splicing of pre-tRNA + OH 5’ P 1. Endo-nuclease 2. Phospho-diesterase 3. Kinase (ATP) 4. Ligase (ATP) 5. Phosphatase 2’,3’ cyclic phosphate Spliced tRNA Intron of 10-20 nucleotides + Excised intron
Splicing of Group I and II introns • Introns in fungal mitochondria, plastids, Tetrahymena pre-rRNA • Group I • Self-splicing • Initiate splicing with a G nucleotide • Uses a phosphoester transfer mechanism • Does not require ATP hydrolysis. • Group II • self-splicing • Initiate splicing with an internal A • Uses a phosphoester transfer mechanism • Does not require ATP hydrolysis
Splicing of pre-mRNA • The introns begin and end with almost invariant sequences: 5’ GU…AG 3’ • Use ATP to assemble a large spliceosome • Mechanism is similar to that of the Group II fungal introns: • Initiate splicing with an internal A • Uses a phosphoester transfer mechanism for splicing