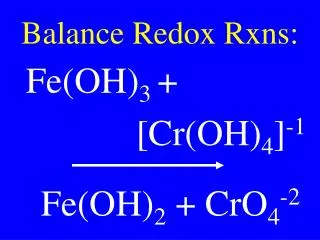Understanding Redox Reactions: Oxidation and Reduction Explained
Redox reactions are fundamental to energy production in various processes, including photosynthesis and combustion. In a redox reaction, electrons are transferred between substances, leading to oxidation (loss of electrons) and reduction (gain of electrons). The oxidation states help track electron transfer in reactions involving elements and compounds. For example, in the reaction 2Na(s) + Cl2(g) -> 2NaCl(s), sodium is oxidized, and chlorine is reduced. Understanding these concepts is essential for comprehending chemical reactions and energy transformations in biological and chemical systems.

Understanding Redox Reactions: Oxidation and Reduction Explained
E N D
Presentation Transcript
My favorite reaction... • 2Na(s) + Cl2(g)->2NaCl(s) • electron is transferred • This is called oxidation-reduction or redox
Redox • Involves most rxns for energy production • photosynthesis, combustion, breakdown of sugars, fats, proteins
Oxidation States • A way to keep track of electrons in redox rxns • Oxidation states of covalent cmpds- arbitrarily assigned to certain atoms
Rules- Oxidation States • Oxidation state of an atom in an element is zero.
Element Examples... • Na(s), O2(g), O3(g), Fe(s), Hg(l) all have an oxidation state of 0
Monatomic Ions • Oxidation state of monatomic ion is the same as its charge
Monatomic Ion Ex • Oxidation state of: • Na+ = +1 • Cl- = -1 • Ca2+ = +2
Fluorine • In its compounds, fluorine always has an oxidation state of -1.
Oxygen • Assigned oxidation state of -2 in its covalent compounds. • CO, CO2, SO2, SO3
Hydrogen Rules • In covalent compounds with nonmetals, H is assigned an oxidation state of +1.
Hydrogen examples • HCl, NH3, H2O, CH4 • all have hydrogen at +1 oxidation state
Sum of the states • Sum of the oxidation states must be 0 for neutral compounds or the charge of the ion for an ion
Examples • Sum of states in CO32- • -2 • Sum of states in NH4+ • +1
Let’s Try It! • CO2 • SF6 • NO3- • K2CrO4
Redox Rxns • Some rxns are characterized by literal e- transfer • 2Na(s) + Cl2(s)->2NaCl(s)
Less obvious redox • CH4(g) + 2O2(g) -->CO2(g) +H2O(g) • Assign oxidation states
A closer look • Oxidation is an increase in oxidation state (a loss of electrons)
A closer look • Reduction is a decrease in oxidation state (a gain of electrons)
In the reaction... • 2Na(s) + Cl2(s)->2NaCl(s) • sodium is oxidized • chlorine is reduced
However... • Chlorine is the oxidizing agent (e- acceptor) • sodium is the reducing agent (e- donor)
In the other rxn... • C is oxidized (lost e-) • O is reduced (gained e- • CH4 is reducing agent • O2 is oxidizing agent