Understanding VSEPR Theory: Predicting Molecular Geometry
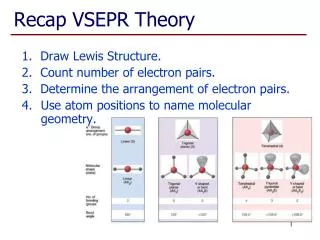
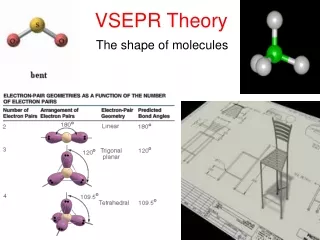
VSEPR Theory (Valence Shell Electron Pair Repulsion) is a model used to predict the shapes of molecules based on the arrangement of electron pairs around a central atom. According to the theory, electron pairs repel each other and thus position themselves as far apart as possible. This results in distinct molecular geometries based on the number of atoms and unshared electron pairs. Examples include linear (BeF2), trigonal planar (AlF3), tetrahedral (CH4), trigonal bipyramidal (PF5), and octahedral (SF6) configurations. Also discussed are bent (H2O) and trigonal pyramidal (NH3) shapes for molecules with lone pairs.

Understanding VSEPR Theory: Predicting Molecular Geometry
E N D
Presentation Transcript
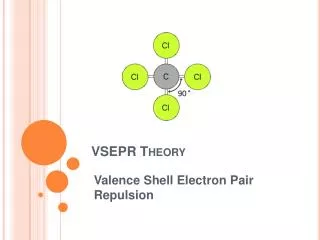
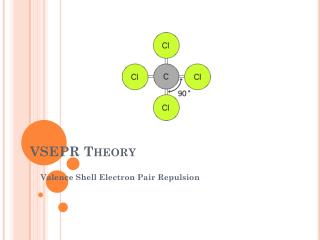
VSEPR Theory Valence Shell Electron Pair Repulsion
VSEPR Theory • Predicts the molecular shape of a bonded molecule • Electrons around the central atom arrange themselves as far apart from each other as possible • Unshared pairs of electrons (lone pairs) on the central atom repel the most • So only look at what is connected to the central atom
These are for molecules with only paired electrons around the central atom.
Linear • 2 atoms attached to center atom • 0 unshared pairs (lone pairs) • Bond angle = 180o • Type: AX2 • Ex. : BeF2
Trigonal Planar • 3 atoms attached to center atom • 0 lone pairs • Bond angle = 120o • Type: AX3 • Ex. : AlF3
Tetrahedral • 4 atoms attached to center atom • 0 lone pairs • Bond angle = 109.5o • Type: AX4 • Ex. : CH4
Trigonal Bipyramidal • 5 atoms attached to center atom • 0 lone pairs • Bond angle = • equatorial -> 120o • axial -> 90o • Type: AX5 • Ex. : PF5
Octahedral • 6 atoms attached to center atom • 0 lone pairs • Bond angle = 90o • Type: AX6 • Ex. : SF6
These are for molecules with both paired and unshared (lone) pairs of electrons around the central atom.
Bent • 2 atoms attached to center atom • 2 lone pairs • Bond angle = 104.5o • Type: AX2E2 • Ex. : H2O
Trigonal Pyramidal • 3 atoms attached to center atom • 1 lone pair • Bond angle = 107o • Type: AX3E • Ex. : NH3