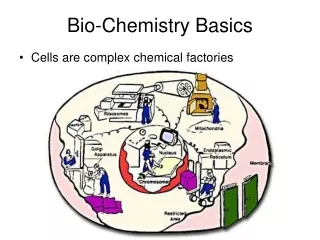
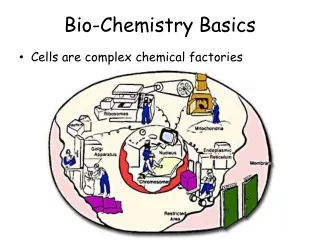
Basics of Chemistry
280 likes | 443 Vues
Basics of Chemistry. Week 4 Dr. Thornton OAPB. The Atom by Niels Bohr. electron (negative charge). electron shell. proton (positive charge). nucleus. neutron (no charge). Hydrogen (H). Helium (He). The Bohr Model. Electron Shells. Elements. Matter: takes up space, has weight.

Basics of Chemistry
E N D
Presentation Transcript
Basics of Chemistry Week 4 Dr. Thornton OAPB
The Atom by Niels Bohr electron (negative charge) electron shell proton (positive charge) nucleus neutron (no charge) Hydrogen (H) Helium (He) The Bohr Model
Elements • Matter: takes up space, has weight. • Mass: the quantity of matter in a given object. • Energy: review the types of energy.
electron transfer sodium ion (Na+) chloride ion (CI–)
ice In ice, the maximum number of hydrogen bonds form, causing the molecules to be spread far apart. liquid water In liquid water, hydrogen bonds constantly break and re-form, enabling a more dense spacing than in ice.
pure water (a) Starting with pure water Pure water is a “neutral” substance in terms of its pH levels. (H2O)
(c) Making water more basic An equal concentration of sodium hydroxide, poured into water, dissociates into Na+ and OH– ions, moving the water toward the basic end of the scale. NaOH base
(d) Combining acidic and basic solutions When the acid and base solutions are poured together, the OH– ions from (c) accept the H+ ions from (b), forming water and keeping the solution at a neutral pH. neutralized solution