Organic Chemistry Basics
This resource explores the essential properties of carbon, its role as a non-metal with four valence electrons, and its ability to form various compounds. Learn the prefixes for naming carbon compounds, including methane to decane, and discover the types of organic compounds such as hydrocarbons and carbohydrates. The guide includes insights into hydrocarbon types (alkanes, alkenes, alkynes), petrochemicals, and the significance of functional groups. It also covers the distinction between saturated and unsaturated hydrocarbons and introduces both natural and synthetic polymers.

Organic Chemistry Basics
E N D
Presentation Transcript
Organic Chemistry Basics General Chemistry
Properties of Carbon • Carbon is a non-metal • Carbon has 4 valence electrons. • Carbon can form up to 4 bonds. • The Lewis Dot Structure for carbon shows how carbon can form these 4 bonds.
Prefixes for Carbon Compounds • Meth – 1 carbon • Eth – 2 carbons • Prop – 3 carbons • But – 4 carbons • Pent – 5 carbons • Hex – 6 carbons • Hept – 7 carbons • Oct – 8 carbons • Non – 9 carbons • Dec – 10 carbons
Types of Organic Compounds • Hydrocarbons – contains the elements, hydrogen and carbon • Carbohydrates – contains the elements, hydrogen, carbon, and oxygen • Complex polymers • Biological molecules
Hydrocarbons • Three basic hydrocarbons • Alkanes – single bonds between carbon atoms • Alkenes – double bonds between carbons atoms • Alkynes – triple bonds between carbon atoms • Examples: • C-C is ethane • C=C is ethene • C= C is ethyne
Petrochemicals • Petrochemicals contain hydrocarbons. • Propane, butane, and octane are some of the most common. • Propane is a single chained carbon molecule with 3 carbon atoms • Butane is a single chained carbon molecule with 4 carbon atoms. • Octane is a single chained carbon molecule with 8 carbon atoms
Identification of Carbon Chains • Draw a Lewis Dot of the molecule. • Identify the number of hydrogen atoms and attach as appropriate. Spread evenly. • Keep functional groups and carbon/hydrogen groupings together. • Name the compound based on Functional Groups.
Name and Draw the Following Compounds • CH4 methane • C2H6 ethane • C2H4 ethene • C2H2 ethyne • CH3CH2OH ethanol • CH2O formaldehyde • C6H6 benzene • CH3COOH acetic acid (ethanoic acid)
Saturated vs. Unsaturated Hydrocarbons Saturated – a molecule whose carbon atoms bond to the maximum number of hydrogen atoms Unsaturated – a molecule that contains a carbon-carbon multiple bond, to which more hydrogen atoms can be added Alkanes are typically saturated. Alkenes and alkynes are unsaturated due to their multiple bonds.
Carbohydrates • Composed of carbon, hydrogen and oxygen. • Examples: • Sucrose • Glucose
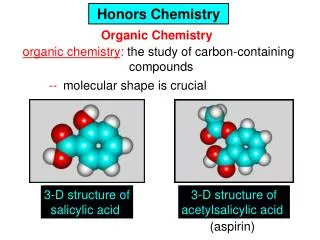
Polymers • Polymers are created when small molecules link together in repetitive subunits. • Polymers can be natural or synthetic (human-made) • Natural polymers: proteins and nucleic acids • Synthetic polymers: polythene, nylon, and Kevlar • Common pharmaceuticals such as aspirin, vitamins, and insulin are organic.
Natural Polymers • Proteins – large biological molecule made of many amino acids linked together though amide (peptide) bonds • Peptide – an amide bond that links two amino acids together • DNA • RNA • Amino Acids – a molecule that contains both an amino group and a carboxylic acid functional group.
Synthetic Polymers • Nylon– a large molecule that is made of repeating units containing polyamide (nitrogen containing functional group) • Kevlar – see reading • Plastics – compounds formed from petrochemicals which consist of long chains. The stronger the plastic the more layering of these long chains. • Why are bottles for soda, much stronger than those of water?