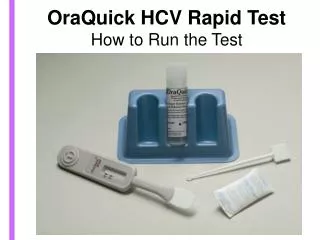
OraQuick HCV Rapid Test How to Run the Test
580 likes | 1.2k Vues
OraQuick HCV Rapid Test How to Run the Test . Overview. Background Presentation D emo of the test You will practice running 2 tests We will not be testing anyone in the room Proficiency Test - You will run 2 tests without assistance R un, read, and document 2 tests

OraQuick HCV Rapid Test How to Run the Test
E N D
Presentation Transcript
Overview • Background Presentation • Demo of the test • You will practice running 2 tests • We will not be testing anyone in the room • Proficiency Test - You will run 2 tests without assistance • Run, read, and document 2 tests • Read results of 10 test images • Pass/fail course, must get 100% to pass
Intro to OraQuick HCV Rapid Test • One-step test • Visual interpretation • Uses blood only • Looks for HCV antibodies • Results in 20 to 40 minutes
Who can run the test? • California Health and Safety Code Section 120917 allows non-medical personnel that have been trained as HIV test counselors to perform CLIA-waived HCV tests if they: (Next Slide)
(continued...) • 1. Have been trained in HIV test counseling by OA or its agents; • 2. Work in a HIV testing site that is funded by OA or that uses staff trained by OA or its agents and has a QA plan approved by the local health department and has HIV testing staff that complies with state regulatory QA requirements. • 3. Have been trained by OA or its agents in both HIV and HCV test kit proficiency for finger-stick blood tests and in universal infection control precautions, consistent with best infection control practices.
Package Inserts • Contains instructions • CLIA requires following the manufacturer’s instructionsto the letter!!
Test Accuracy • We’re going to talk about how well the test works • You do not need to memorize what I’m saying - only need to know test is very accurate • There are two components of test accuracy: • Sensitivity • Specificity
Specificity • When a test finds something, it should be the right thing • Tests ability to detect a true negative
Specificity Not exact, but very accurate
Sensitivity • The ability of a test to find what it’s looking for and not miss anything. • Test’s ability to detect a true positive
Sensitivity The test’s ability to detect true positives and not miss anything HCV test
Combining Specificity and Sensitivity An ideal test would find the right thing (specificity) and not miss anything (sensitivity)
Think of a Tuna net…. A net that gets all the tuna and none of the dolphins
Because the Test is Highly Sensitive… • We do not need to confirm negatives • We do, however, always need to do a follow-up test to find out if the client has HCV now
Testing Process for HCV OraQuick Test Negative Reactive Client referred to follow-up HCV nucleic acid testing
With Clients… • Emphasize that the test is extremely accurate • De-emphasize statistics and percentages • For example “This test is highly accurate”
Quality AssuranceRequirements • QA are practices and procedures which ensure that every client receives an accurate test result • QA reduces human error as much as possible
Components of Quality Assurance • Personal and logistical characteristics • External controls • Lab space • Universal precautions
Must Have… Steady hand Good eye sight Organizational skills Adequate lighting • Full/bright light, task light • Do not use a flashlight
External Controls • Fluids made from human plasma • Biohazards – wear gloves! • Negative, Positive • Store controls at 36 to 46 degrees • Expire 56 days after opening
External Controls are Used for: • Training, we will use them today • Determining if test is working properly • Determining if lighting is adequate • Determining if the test reader has sufficient eye site
When to Run External Controls • New operator • New setting or conditions changed significantly • New test kit lot or shipment • Out of range testing area or storage area temperature • Two invalid results in a row* • Every 40 tests or once a month (whichever comes first)
Temperature Control • Perform test, • 59-99 degrees • If testing temperature is out of required temperature range stop testing • If out of temp range, run controls before proceeding • Store test: 36-86 degrees
Lab Space • A space for undisturbed test processing • Your site is a lab • Separate from counseling area • No smoking, eating or drinking
Universal Precautions The universal practice of avoiding contact with patients' bodily fluids, (blood) by means of the wearing of nonporous articles such as medical gloves
Gloves • Wear them when handling blood or blood products, and…. • TODAY– whenever you touch the control fluid vials!!!! • How often do • you change them? • How do you remove them?
Sharps: Handling & Disposal • Medical instruments that are used to puncture the skin (syringes, lancets, needles) • Dispose of sharps immediately, in a hard redplastic bio bin! • Do NOT dispose of sharps in a red bio hazard bag
Biohazard Bags Handling & Disposal • Bandages, used cotton and gauze, and gloves with body fluids on them are bio hazardous waste • By law, if fluid cannot be squeezed out of the cotton, gauze, etc., the waste item can be disposed of in regular trash
For Today’s Training • Place loops and used test kits in the sharps container • Place gloves and all other trash in the brown paper bag
Paperwork Needed • Expanded Checklist • Short Checklist (Competency Assessment Test list) • Rapid testing log or lab slip • Lab stickers • HIV/HCV Testing Form • Always use blue or black ink only!
Test developer solution and vial Reusable test stand Specimen collection loop Result window Test device Flat pad Absorbent packet
Test Device Control line Test line Flat pad Vent holes – Do NOT obstruct Paddle must be inserted into the vial no later than 60 minutes after sample has been introduced
Trainers Demonstrate • Trainer Reads • Other Trainer preforms test • Participants follow along (Expanded Checklist)
Participants’ Practice • No food or drink • Listen to detailed steps: • Only do what we tell you to do • Do all steps in order • If you finish a step quickly, wait for the next step
Reading Time • Results in 20-40 minutes • If a reactive result appears before 20 minutes have passed, the result may still not be read until at least 20 minutes have passed
OraQuick Test Results • Two lines: • “C” - Control line • “T” - Test line • Negative result • Reactive result • Invalid result
What causes invalids? • Human error (e.g., no specimen) • Unknown • Manufacturer error If you ever have an unusual result, do not deliver it
What does it mean if a client has an invalid test result?What does the darkness of the lines mean? NOTHING
A D B C C C C C T T T T G H F E C C C C T T T T
Internal Control – The “C” Line • The control line is the “C” line • Internal control tells us: • Specimen was adequately applied • Proper hydration • Migration of reagents past the “T” zone. Internal & external controls are standard lab practice – not a sign of test kit unreliability
Test Line – The “T” Line • The “T” line is the “test line”. It works with the external control fluids to: • tell us if the result is reactive or non-reactive. • tell us if the reader can see lines. • tell us if there is proper lighting. • tell us if the reader’s eyesight is adequate to run the test.
Controls Work Together • If the internal control & the external controls both tell us the test kit is working, why do we need both? • Because they tell us something different! • “C” line tells us test kit is working properly • “T” line tells us the test kit can detect HCV antibodies when they’re present
Results of Practice Test • Did you get the correct result? • Any questions? • The control fluid that we use provides a “challenge sample” • Light control line verifies lighting is OK • Verifies test kit detecting small amount of antibody • Darkness of line NOT related to viral load, disease progression, or anything else about the client
If a Client Asks to See the Test Kit, What Would You Do? • Say “No” – Why? • Could compromise confidentiality • Only trained personnel may read the test • Test is disposed of in biohazard bag as soon as it’s read • Think about the picture with the pink background – if you have a light line would you be able to see it on this test?
How Do You Correct a Written Mistake? • Use blue or black ink only on all forms • A single crossed-out line, corrected entry written clearly above, include date and initials of the individual making the change. • At no time should an original entry be obliterated or otherwise made illegible by a change on the record. 7:22pm T.K. 12/15/2011 End time: 7:12pm
Any questions? Proficiency tests coming next….
OraQuick HCV Test Proficiency Words of Caution • Follow the checklist!!!! • This is not a test of memorization • Run 2 tests using 2 differentcontrolvials • Put your name on the top of your paper(s) • Take your time, double & triple checking your work • If you make a mistake on your paperwork • and you catch it, you can fix it. • and I catch it, you will have to come back another day