Horse chestnut seed for chronic venous insufficiency
Michelle Orengo-McFarlane, MD University of California at San Francisco Family and Community Medicine. Horse chestnut seed for chronic venous insufficiency. Chronic venous insufficiency (CVI) affects 10-15% of men and 20-25% of women

Horse chestnut seed for chronic venous insufficiency
E N D
Presentation Transcript
Michelle Orengo-McFarlane, MD University of California at San Francisco Family and Community Medicine Horse chestnut seed for chronic venous insufficiency
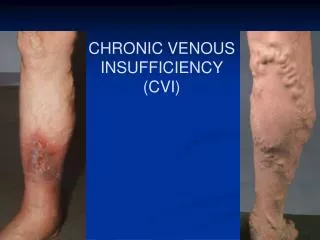
Chronic venous insufficiency (CVI) affects 10-15% of men and 20-25% of women Characterised by chronic inadequate drainage of venous blood and venous hypertension, which results in leg edema (swelling) dermatosclerosis (hardening of the skin) Feelings of pain, fatigue and tenseness in the lower extremities May be complicated by skin ulceration chronic (and potentially life-threatening) infections of the lower extremities Introduction
Conservative therapy consists largely of compression treatment This often causes discomfort and has been associated with poor compliance Oral drug treatment is an attractive option, but no drugs in the standard US pharmacopoeia provide this Introduction
Native to the Balkan Peninsula Grown throughout the northern hemisphere Related to Aesculus californica, the California buckeye Traditionally used in European herbal medicine for circulatory disorders and ulcer treatment The “most widely prescribed oral antiedema venous remedy in Germany” Horse chestnut (Aesculus hippocastanum)
To “review the evidence from rigorous clinical trials assessing the efficacy and safety of HCSE versus placebo, or reference therapy, for the symptomatic treatment of CVI.” Goals of meta-analysis
Inclusion criteria: Randomised controlled trials (RCTs) Study participants = patients with CVI Oral preparations containing HCSE as the only active component (mono-preparation) compared to placebo or reference therapy HCSE was standardised to escin which is the primary active constituent Clinical outcome measures: CVI-related symptoms (leg pain, pruritus, edema), leg volume, circumference at ankle and calf, and adverse events Methods
2 of the authors (Pittler and Ernst) selected trials and extracted data Quality of studies was assessed using the Jadad scale, giving each study a score of 1-5 (higher scores indicate better study quality) Concealment of treatment allocation was scored: A = clearly concealed, B = unclear if concealed, C = clearly not concealed, D = concealment of allocation was not used. Only RCTs were included Methods
29 randomized controlled clinical trials were identified, and 17 trials met inclusion criteria All RCTs except one were doubleblinded All scored at least one out of five points on the Jadad scale 8 good quality 5 intermediate 3 poor 3 trials scored A and 14 trials scored B for the method of allocation concealment Results
Dose range: 100-150mg escin 15 100mg daily (50 BID) 2 150mg daily (75 BID) Duration: 2-16 weeks Most 3-8 weeks Results
10 were placebo controlled 2 compared HCSE with compression stockings + placebo 5 were controlled against reference medication 4 = O-ß-hydroxyethyl rutosides (HR) - a plant-derived flavonoid 1 = pycnogenol – a flavonoid-containing water extract of the bark of the French maritime pine Results
Seven placebo-controlled trials Six studies (n = 543) reported a statistically significant reduction (P < 0.05) of leg pain compared with placebo One reported an improvement compared with baseline Studies which compared HCSE with HR, pycnogenol or compression reported no significant difference Results: leg pain
Six placebo-controlled trials Five trials (n = 491) reported a statistically significant reduction of edema compared with placebo Results: edema
One study reported that HCSE was inferior to pycnogenol One reported no significant differences compared with compression Results: edema
8 placebo-controlled trials 4 trials (n = 407) suggested a statistically significant reduction of pruritus compared with placebo (P < 0.05) One found no significant difference compared with compression Results: pruritis
7 placebo-controlled trials All of these studies used water displacement plethysmometry to measure this outcome Meta-analysis of 6 (n = 502) suggested a WMD of 32.1ml (95% CI 13.49 to 50.72) vs placebo (pooled standardised mean difference 0.34; 95% CI 0.15 to 0.52) One suggested HCSE was not significantly different then HR One suggested that it may be as efficacious as treatment with compression stockings (WMD -2.90 ml; 95% CI -30.42 to 24.62) Results: leg volume
3 studies, all using 100 mg escin daily, reported a statistically significant reduction of mean leg volume after two weeks of tx vs placebo (P < 0.01) Persistence of treatment effects was suggested by one study - at the end of a six week follow-up period, mean leg volume was similar to post-treatment values Results: leg volume
7 placebo controlled trials 5 (n = 172) suggested a statistically significant reduction at the ankle, 3 (n = 112) at the calf vs placebo Ankle: meta-analysis of 3 trials w/ adequate data suggested a statistically significant reduction vs placebo (WMD 4.71 mm; 95% CI 1.13 to 8.28; pooled standardised mean difference 0.60; 95% CI 0.15 to 1.05). Calf: pooled analysis of three trials suggested a statistically significant reduction vs placebo (WMD 3.51 mm; 95% CI 0.58 to 6.45; pooled standardised mean difference 0.42; 95% CI -0.04 to 0.88). Results: limb circumference
14 studies 4 reported no treatment-related adverse events 6 reported GI complaints, dizziness, nausea, headache and pruritus; frequency 1-36% of treated patients. 4 reported good tolerability Results: adverse effects
Limited data on adherence - only 3 trials reported that adherence was monitored Most studies had a small sample size Drop-out rates ranged 0-19.5% Insufficient data on effect size top assess heterogeneity for most studies, though the data on leg volume appear fairly homogenous Limitations
Did effect vary by quality of studies? – was not evaluated Were all relevant studies included? - unknown Evidence of unreported negative studies - not evaluated Was effect size clinically significant? Objective measures were significantly positive, though there was limited data on effect size for subjective outcomes Limitations
HCSE appears to be a safe and well-tolerated treatment for CVI Objective measures were significantly positive and most studies were good quality More data is needed to establish long-term safety and efficacy Caution is advised in recommending any US-manufactured supplement, as they are minimally regulated under DSHEA Identify reputable supplement companies Discussion
I would recommend this to my patients. The bottom line
A Modern Herbal: http://www.botanical.com/botanical/mgmh/mgmh.html NCCAM on HCSE: http://nccam.nih.gov/health/horsechestnut/ DSHEA: http://www.cfsan.fda.gov/~dms/ds-oview.html Schulz V, et al. Rational Phytotherapy. Berlin: Springer-Verlag; 1998:129-138. Image credit: http://arboreality.blogspot.com/2007/06/horse-chestnut-tree-in-spring.html References