Exploring Chemical Kinetics: Rates, Mechanisms, and Experiments
Delve into Chemical Kinetics to understand reaction rates, mechanisms, and experimentally derived rate laws. Learn how collisions and reaction steps influence reaction rates and discover the interplay of catalysts and intermediates.

Exploring Chemical Kinetics: Rates, Mechanisms, and Experiments
E N D
Presentation Transcript
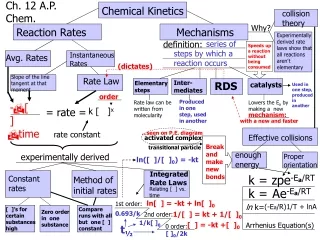
Ch. 12 A.P. Chem. Chemical Kinetics collision theory Why? Reaction Rates Mechanisms Experimentally derived rate laws show that all reactions aren’t elementary series of steps by which a reaction occurs definition: Speeds up a reaction without being consumed Instantaneous Rates Avg. Rates (dictates) Slope of the line tangent at that moment Rate Law Inter-mediates RDS Elementary steps catalysts Used in one step, produced in another order Produced in one step, used in another Rate law can be written from molecularity Lowers the Ea by making a new ______________ -D[ ] = rate = k [ ]x mechanism: Dtime with a new and faster rate constant ...seen on P.E. diagram Effective collisions activated complex Break and make new bonds transitional particle enough energy experimentally derived Proper orientation ln([ ]/[ ]0) = -kt Integrated Rate Laws k = zpe-Ea/RT Constant rates Method of initial rates Relating [ ] vs. time k = Ae-Ea/RT ln[ ] = -kt + ln[ ]0 1st order: ln k= (-Ea/R)1/T + lnA [ ]’s for certain substances high Compare runs with all but one [ ] constant Zero order in one substance 0.693/k 1/[ ] = kt + 1/[ ]0 2nd order: 1/k[ ]0 [ ] = -kt +[ ]0 Arrhenius Equation(s) 0 order: t½ [ ]0/2k
Rate1= 3.4x10-8 = k[5.1x10-4]m[0.35x10-4]n Ch. 12 Review Problem 1= =0.34m Rate4= 3.4x10-8 = k[1.5x10-3]m[0.35x10-4]n m= 0 The transfer of an O atom from NO2 to CO has been studied at 480 K, CO(g) + NO2(g) ----> CO2(g) + NO(g) and the following data was collected, ---initial conc. (mol/L)--- initial rate (mol L-1 hr-1) [CO] [NO2] #1) 5.1 x10-4 0.35 x 10-4 3.4 x 10-8 #2) 5.1 x10-4 0.70 x 10-4 1.36 x 10-7 #3) 5.1 x10-4 0.18 x 10-4 8.5 x 10-8 #4) 1.5 x10-3 0.35 x 10-4 3.4 x 10-8 A. Write the generic rate law for this reaction. B. Find the order of the reaction with respect to each reactant using the data provided. C. Calculate the rate constant from the data provided. D. Find the half-life for the reaction for run #1 above. F. Which mechanism below best agrees with the experimentally derived rate law: #1 NO2 + CO --> CO2 + NO #2 (slow) NO2 + NO2-->NO3 + NO (fast) NO3 + CO-->NO2 + CO2 #3 (slow) NO2 --> NO +O (fast) CO + O --> CO2 1st order in NO2 and CO Therefore n=2 Doubles ↓ Same ↓ Quadruples ↓ 2nd order in NO2 and zero order in CO OK 1st order in NO2 and zero order in CO Rate = k [CO]m [NO2]n Rate = k [NO2]2 3.4 x 10-8 mol L-1hr-1 = k [0.35 x 10-4 mol/L]2 k = 28 L/mol·hr t½= 1/k[NO2]0 =1.0x103 hr
2C4H10 + 13O2 → 8CO2 + 10 H2O -D[C4H10] Dtime Are these equal? -D[ O2 ] Dtime
2C4H10 + 13O2 → 8CO2 + 10 H2O -6.5D[C4H10] = Dtime Are these equal? -D[ O2 ] Dtime