Matter, Elements, Compounds and Mixtures
Matter, Elements, Compounds and Mixtures. Grand Prismatic Spring in Yellowstone National Park. Source: David Maisel/Stone/Getty Images. Why does a hot dog get hot in the microwave? Why does wood burn but rocks do not? How does soap work? When Iron rusts, what is happening to it?

Matter, Elements, Compounds and Mixtures
E N D
Presentation Transcript
Grand Prismatic Spring in Yellowstone National Park Source:David Maisel/Stone/Getty Images
Why does a hot dog get hot in the microwave? • Why does wood burn but rocks do not? • How does soap work? • When Iron rusts, what is happening to it? • Why doesn’t Aluminum rust?
How does this lush vegetation grow in a tropical rain forest, and why is it green?
These are all questions that can and will be answered by studying Chemistry • In this chapter we will look at the nature of matter, how it is organized, why it changes, and the overriding Law that governs these changes
Characteristics of Matter 1. What are the 2 characteristics of matter? • matter is anything that has mass and occupies space 2. There are three ways of describing matter, what are they? • Volume, mass, weight
3. Of these three ways, which one is the most accurate for describing matter? • mass 4. Why are volume and weight not reliable ways of describing matter? • Volume can change with temperature and weight can change with gravity
Be able to: 1. Define matter and its three states and identify its properties
States of Matter 5. What is the easiest way of subdividing matter? • By their phases 6. What are the three phases of matters • Solid, Liquid and Gas 7. Differentiate between the three phases of matter
Be able to: 2. Learn to distinguish between physical and chemical properties and changes and be able to identify them
Physical and Chemical Properties 8. When we refer to the properties of matter, what do we mean by that ? • Properties are a set of characteristics which we use to identify a substance 9. How many types of properties are there? • Physical-what can be observed by just examining it without the production of a new substance • Chemical-describes how the substance interacts with other substances to produce a new substance.
10. What are some examples of physical and chemical properties?
An iron pyrite crystal (gold color) on a white quartz crystal.
Examples of Physical Properties Is it a gas, liquid, or solid?
Gallium metal has such a low melting point (30°C) that it melts from the heat of a hand.
Examples of Physical Properties taste odor length volume density
Examples of Chemical Properties How a chemical reacts with another chemical is a chemical property
Examples of Chemical Properties How a chemical reacts with another chemical is a chemical property
Examples of Chemical Properties Reaction with water to produce new substances
Examples of Chemical Properties How a substance reacts in the presence of heat
Chemical vs Physical Changes • all reactions in chemistry are either physical or chemical changes • by understanding what kind of change occurred we can better understand the reaction 11. What is the difference between a physical and a chemical change?
Chemical vs Physical Changes • Physical changes involved a change in odor, size, color, phase but not a change in what the substance is. • Chemical changes always results in the production of a new substance. What the substance once was, it no longer is...
Chemical v Physical Changes? • the components were mixed in the liquid. The chromatography process simply separates the mixture into its individual components without changing what they are
Chemical Changes Chemical changes always results in the production of a new material
Chemical v Physical Changes? Production of new materials: Chemical Change!
Chemical v Physical Changes? • What is produced now has a new mass therefore it has added something from the air to its chemical makeup. A new substance: Chemical
12. What is the difference between a physical change for water and a chemical change?
Add or take away heat, water changes phase but not what it is
Run electricity through water(electrolysis) and water breaks down into Hydrogen gas and Oxygen gas
Let’s Practice! 1. Classify each of the following as a physical or a chemical property • The boiling point of a certain alcohol is 78ºC • Diamond is very hard • Sugar ferments to form alcohol • A metal wire conducts an electric current
2. Classify each of the following as a physical or a chemical change A. Iron is melted B. Iron combines with oxygen to form rust C. Wood burns in air D. A rock is broken into small pieces
3. Classify each of the following as a chemical or a physical change or a combination of the two A. Milk turns sour B. Wax is melted over a flame and then catches fire and burns
Be Able To: 3. Distinguish between elements, compounds, and mixtures and be able to identify them
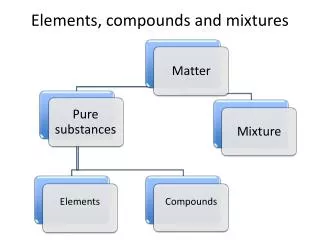
Classification Scheme of Matter Matter Mixtures Pure Substances Homogeneous Heterogeneous Elements Compounds
Pure substances 13. What is a pure substance? • Kind of matter whose samples have the same properties or characteristics • always made up of the same “stuff”
Pure substances 14. What are the two kinds of pure substances? • elements and compounds
Pure substances • Pure substances are will have the same consistency no matter how closely you look at it.
Pure substances • Even at the atomic level as this electron micrograph shows
Elements 15. What are elements? • substances that cannot be broken down into any other by ordinary chemicalmeans. Nuclear reactions have been able to convert one atom into another
Elements 16. Name some examples of elements? • Sulfur • Oxygen • Carbon • Sodium • Potassium • Iron
Compounds 17. What are compounds? • Matter made up of more than one type of element, can be broken down into elements through chemical means
18. Is there any order to the way these elements form compounds? • the elements that make up the compound are always found in the same whole number ratio
19. What are examples of compounds? • NaCl(always a 1 Na : 1 Cl ratio) • H2O(all water in the universe has a • 2H : 1O ratio) 20. How does the chemical and physical properties of a compound compare with the properties of the elements that make it up?