Elements, Compounds, and Mixtures
240 likes | 732 Vues
Explore the characteristics of elements, pure substances, and their identification methods in this informative guide. Learn about the unique properties of metals, non-metals, and metalloids. Test your knowledge with engaging mini-quizzes!

Elements, Compounds, and Mixtures
E N D
Presentation Transcript
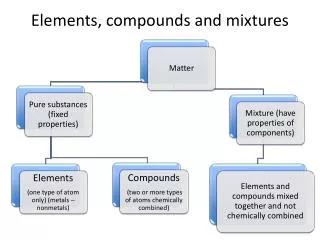
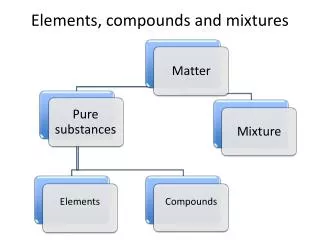
Elements, Compounds, and Mixtures Elements
Essential Questions • What are pure substances? • What are the characteristics of elements? • How can they be used to identify an element?
Elements • Are a pure substance • Can NOT be broken into a simpler substance physically or chemically
Pure substances • Have only one type of particle • Particles are called atoms • All atoms are EXACTLY the same
Characteristic Properties • Characteristics do not depend on the amount of the element • Physical characteristics: • Boiling point • Melting point • Density,
Characteristic Properties • Characteristics do not depend on the amount of the element • Chemical characteristics: • Reactivity with acid • Flammability
Identifying Elements • Elements are unique. • Elements may share some properties with other elements, but not ALL properties.
Identifying Elements • Three major categories of elements: • Metals • Non-metals • Metalloids
Metals • Shiny • Conduct heat energy • Conduct electric current • Malleable • Ductile
Nonmetals • Do not conduct heat energy • Do not conduct electric current • Solid nonmetals have a dull appearance. • Brittle • Unmalleable • Examples: Iodine, sulfur, neon
Metalloids • Have properties of both metals and nonmetals • Are also called “semiconductors” • Some are shiny. • Some are dull. • Are somewhat malleable and ductile • Ex: Silicon, boron, antimony
Mini Quiz 1. A sample of matter, either a single element or a single compound, that has definite chemical and physical properties is a(n) A. Metal B. Element C. Metalloid D. Pure Substance
Mini Quiz 4. Elements are classified in 3 major categories. Which of the following does not apply? A. Metal B. Non-metal C. Metalloid D. Compound
Mini Quiz 2. A property of an element that does not depend on the amount of the element is called a(n) • a. electromagnetic property. • b. finite property. • c. unique property. • d. characteristic property.
Mini Quiz 3. An element’s ability to react with acid is an example of a • a. pure substance. • b. physical property. • c. chemical property. • d. melting point.
Reference Holt, Rinehart and Winston (2005) INSB 0-03-043217-0