Elements, Compounds, and Mixtures
Elements, Compounds, and Mixtures. 7E Classify samples of matter from everyday life as being elements, compounds or mixtures. CLASSIFICATION OF MATTER. Pure Substance. Has a definite composition Examples: elements – rock salt compounds – sand or silicon dioxide. Elements.

Elements, Compounds, and Mixtures
E N D
Presentation Transcript
Elements, Compounds, and Mixtures 7E Classify samples of matter from everyday life as being elements, compounds or mixtures.
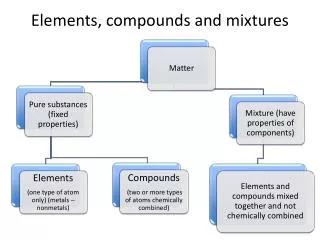
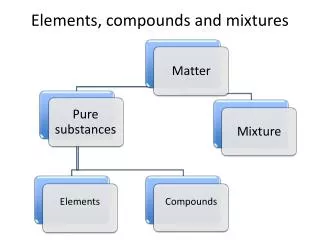
Pure Substance • Has a definite composition • Examples: elements – rock salt compounds – sand or silicon dioxide
Elements • Definite composition • Made of one kind of atom • Cannot be broken down by chemical or physical means • Listed on the Periodic Table
Compound • Definite composition • Made of two or more elements that have been chemically combined • Can be broken down by chemical means such as electrolysis, heat • Example: sodium bicarbonate
Mixture • Variable composition • Two or more pure substances physically mixed together • Can be separated by physical means.
Homogeneous Mixture • Variable composition • Uniformly mixed • Can be separated by distillation or chromatography
Heterogeneous Mixture • Variable composition • Non-uniformly mixed
Separation of Mixtures • Mixtures can be separated by physical means such as distillation; chromatography, filtration, and evaporation • Magnetism can also be used to separate mixtures
Evaporation • A liquid can be removed from a mixture of a solid and liquid by evaporation.
Filtration • A mixture of a solid and liquid can be separated by filtration.
Chromatography • Chromatographycan be used on a larger scale to separate out pure samples from a mixture.
Fractional Distillation • Fractional distillation • Works with liquids with different boiling points. • Only works if all the liquids in the mixture are miscible (e.g. alcohol/water, crude oil etc.)
Simple Distillation • Simple distillation • Separate a pure liquid from dissolved solid impurities which have a very high boiling point.
1. The picture shows a model of the element — • fluorine • helium • beryllium • oxygen
The first equation represents photosynthesis. Plants use energy from sunlight to produce sugar and oxygen from carbon dioxide and water. The second equation represents aerobic respiration. Plants and animals release stored energy in a reaction between sugar molecules and oxygen. This reaction produces carbon dioxide and water. 2. Oxygen (O2) is an example of — • an alloy • a molecule • a salt • a mixture
3. An unknown silvery powder has a constant melting point and does not chemically or physically separate into other substances. The unknown substance can be classified as — • an element • a compound • a mixture • an alloy
4. Which of these are composed of two or more different substances that are chemically combined in a definite ratio? • Compounds • Mixtures • Elements • Solutions
5. Salt is added to a beaker of water and stirred until it is completely dissolved. The salt in this mixture can be separated by — • Chromatography • chemical means only • passing the water through filter paper • allowing the water to evaporate slowly
6. When copper and oxygen chemically unite, they form — • an ion • a gas • a compound • a mixture