NUCLEAR CHEM
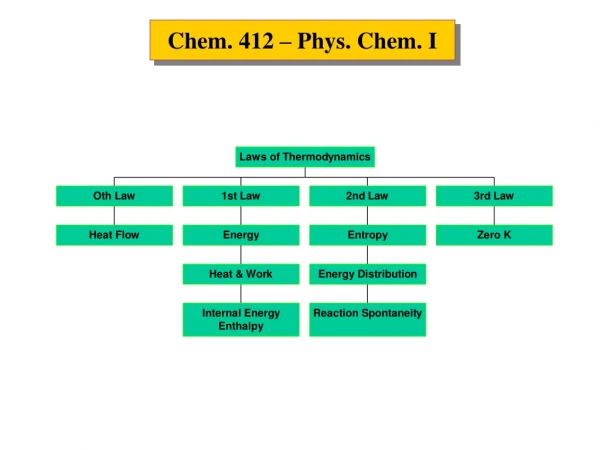
NUCLEAR CHEM. COMPARISON OF CHEMICAL AND NUCLEAR REACTIONS. THREE TYPES OF RADIOACTIVE EMISSIONS IN AN ELECTRIC FIELD. TYPES OF RADIOACTIVE DECAY: BALANCING NUCLEAR EQUATIONS. Alpha decay - A decreases by 4 and Z decreases by 2. Every element heavier than Pb undergoes a decay.

NUCLEAR CHEM
E N D
Presentation Transcript
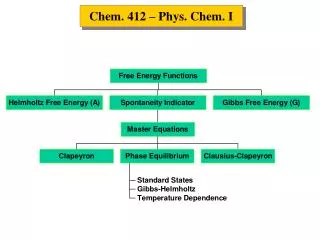
TYPES OF RADIOACTIVE DECAY: BALANCING NUCLEAR EQUATIONS Alpha decay- A decreases by 4 and Z decreases by 2. Every element heavier than Pb undergoes a decay. Beta decay- ejection of a b particle from the nucleus from the conversion of a neutron into a proton and the expulsion of 0-1b. The product nuclide will have the same A but will be one atomic number higher. Positron decay- a positron (0-1b) is the antiparticle of an electron. A proton in the nucleus is converted into a neutron with the expulsion of the positron. A remains the same but the atomic number decreases. Electron capture- a nuclear proton is converted into a neutron by the capture of an electron. A remains the same but the atomic number decreases. Gamma emission- energy release; no change in Z or A.
(a) 23290Th 22888X + 42α 23290Th 22888Ra + 42α (b) 8640Zr + 0-1e AZX 8640Zr + 0-1e 8639Y WRITING EQUATIONS FOR NUCLEAR REACTIONS Write balanced equations for the following nuclear reactions: (a) Naturally occurring thorium-232 undergoes a decay. (b) Zirconium-86 undergoes electron capture. Write a skeleton equation; balance the number of neutrons and charges; solve for the unknown nuclide. A = 228 and Z = 88 A = 86 and Z = 39
A PLOT OF NUMBER OF NEUTRONS vs. NUMBER OF PROTONS FOR THE STABLE NUCLIDES.
NUCLEAR STABILITY AND MODE OF DECAY • Very few stable nuclides exist with N/Z < 1 • The N/Z ratio of stable nuclides gradually increases a Z increases. • All nuclides with Z > 83 are unstable • Elements with an even Z usually have a larger number of stable nuclides than elements with an odd Z • Well over half the stable nuclides have both even N and even Z
PREDICTING THE MODE OF DECAY • Neutron-rich nuclides undergo b decay. • Neutron-poor nuclides undergo positron decay or electron capture. • Heavy nuclides undergo a decay.
PREDICTING NUCLEAR STABILITY Which of the following nuclides would you predict to be stable and which radioactive? Explain. (a)1810Ne (b)3216S (c)23690Th (d)12356Ba Stability will depend upon the N/Z ratio, the value of Z, the value of stable N/Z nuclei, and whether N and Z are even or odd.
Predicting Nuclear Stability Which of the following nuclides would you predict to be stable and which radioactive? Explain. (a)1810Ne (b)3216S (c)23690Th (d)12356Ba PLAN: Stability will depend upon the N/Z ratio, the value of Z, the value of stable N/Z nuclei, and whether N and Z are even or odd. SOLUTION: (a) Radioactive (b) Stable N/Z = 0.8; there are too few neutrons to be stable. N/Z = 1.0; Z < 20 and N and Z are even. (d) Radioactive (c) Radioactive N/Z = 1.20; Figure 23.2A shows stability when N/Z ≥ 1.3. Every nuclide with Z > 83 is radioactive.
PROBLEM: Use the atomic mass of the element to predict the mode(s) of decay of the following radioactive nuclides: Predicting the Mode of Nuclear Decay (a)125B (b)23492U (d)12757La (c)8133As PLAN: Find the N/Z ratio and compare it to the band stability. Then predict which of the modes of decay will give a ratio closer to the band.
PROBLEM: Use the atomic mass of the element to predict the mode(s) of decay of the following radioactive nuclides: (a)N/Z = 1.4 which is high. The nuclide will probably undergo b- decay altering Z to 6 and lowering the ratio. Predicting the Mode of Nuclear Decay (a)125B (b)23492U (d)12757La (c)8133As PLAN: Find the N/Z ratio and compare it to the band stability. Then predict which of the modes of decay will give a ratio closer to the band. SOLUTION: (b) The large number of neutrons makes this a good candidate for a decay. (c)N/Z = 1.24 which is in the band of stability. It will probably undergo b- decay or positron emission. (d)N/Z = 1.23 which is too low for this area of the band. It can increase Z by positron emission or electron capture.
The 238U decay series. Figure 23.3
DN Dt Decay rate (A) = - SI unit of decay is the becquerel (Bq) = 1 d/s. curie (Ci) = number of nuclei disintegrating each second in 1 g of radium-226 = 3.70 x 1010 d/s Nuclear decay is a first-order rate process. Large k means a short half-life and vice versa.
Decrease in the number of 14C nuclei over time. Figure 23.4
PROBLEM: Strontium-90 is a radioactive byproduct of nuclear reactors that behaves biologically like calcium, the element above it in Group 2A(2). When 90Sr is ingested by mammals, it is found in their milk and eventually in the bones of those drinking the milk. If a sample of 90Sr has an activity of 1.2 x 1012 d/s, what are the activity and the fraction of nuclei that have decayed after 59 yr (t1/2 of 90Sr = 29 yr). 2 A0 N0 0.693 k At Nt 29 yr (1.2 x1012 - 2.9 x 1011) Fraction decayed Fraction decayed = = (1.2 x 1012) 0.76 Sample Problem 23.4 Finding the Number of Radioactive Nuclei PLAN: The fraction of nuclei that have decayed is the change in the number of nuclei, expressed as a fraction of the starting number. The activity of the sample (A) is proportional to the number of nuclei (N). We are given the A0 and can find At from the integrated form of the first-order rate equation. SOLUTION: t1/2 = ln so k = = 0.024 yr -1 ln = ln = kt ln At = -kt + ln A0 ln At = -(0.024 yr -1)(59 yr) + ln(1.2 x 1012 d/s) ln At = 26.4 At = 2.9 x 1011 d/s
PROBLEM: The charred bones of a sloth in a cave in Chile represent the earliest evidence of human presence in the southern tip of South America. A sample of the bone has a specific activity of 5.22 disintegrations per minute per gram of carbon (d/min•g). If the 12C/14C ratio for living organisms results in a specific activity of 15.3 d/min•g, how old are the bones (t1/2 of 14C = 5730 yr)? 1 1 A0 15.3 ln 2 0.693 5.22 k At 1.21 x 10-4 yr -1 5730 yr t1/2 PLAN: Calculate the rate constant using the given half-life. Then use the first-order rate equation to find the age of the bones. Sample Problem 23.5 Applying Radiocarbon Dating SOLUTION: k = = = 1.21 x 10-4 yr -1 t = ln = ln = 8.89 x 103 yr The bones are about 8900 years old.
Figure 23.5 Schematic of a linear accelerator.
Schematic of a cyclotron accelerator. Figure 23.6
Figure 23.7 Penetrating power of radioactive emissions. Nuclear changes cause chemical changes in surrounding matter by excitation and ionization. Penetrating power is inversely related to the mass and charge of the emission.
Figure 23.8 The use of radioisotopes to image the thyroid gland. Figure 23.9 PET and brain activity.
The increased shelf life of irradiated food. Figure 23.10
DE c2 The Interconversion of Mass and Energy The mass of the nucleus is less than the combined masses of its nucleons. The mass decrease that occurs when nucleons are united into a nucleus is called the mass defect. E = mc2 DE = Dmc2 Dm = The mass defect (Dm) can be used to calculate the nuclear binding energyin MeV (mega-electron volts). 1 amu = 931.5 x 106 eV = 931.5 MeV
PROBLEM: Iron-56 is an extremely stable nuclide. Compute the binding energy per nucleon for 56Fe and compare it with that for 12C (mass of 56Fe atom = 55.934939 amu; mass of 1H atom = 1.007825 amu; mass of neutron = 1.008665 amu). (0.52846 amu)(931.5 MeV/amu) 56 nucleons Sample Problem 23.6 Calculating the Binding Energy per Nucleon PLAN: Find the mass defect, Dm; multiply that by the MeV equivalent and divide by the number of nucleons. SOLUTION: Mass Defect = [(26 x 1.007825 amu) + (30 x 1.008665 amu)] - 55.934939 Dm = 0.52846 amu Binding energy = = 8.790 MeV/nucleon 12C has a binding energy of 7.680 MeV/nucleon, so 56Fe is more stable.
The variation in binding energy per nucleon. Figure 23.11
Induced fission of 235U. Figure 23.12
A chainreaction of 235U. Figure 23.13
A light-water nuclear reactor. Figure 23.14
Figure 23.15 The tokamak design for magnetic containment of a fusion plasma.