Understanding Oxidation and Reduction in Redox Reactions
In this lesson, we will explore the concepts of oxidation and reduction through the reaction between copper(II) oxide (CuO) and hydrogen (H2) to form copper (Cu) and water (H2O). We will identify what is being oxidized and what is being reduced by balancing half-reactions and understanding electron transfer. Students will practice writing half-reactions, determining which species gains or loses electrons, and will learn about voltaic cells, where these redox reactions can generate electrical current.

Understanding Oxidation and Reduction in Redox Reactions
E N D
Presentation Transcript
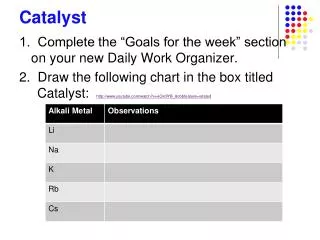
Chem Catalyst Determine what is being oxidized and what is being reduced in the following reaction: CuO + H2 Cu + H2O
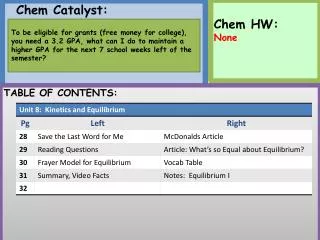
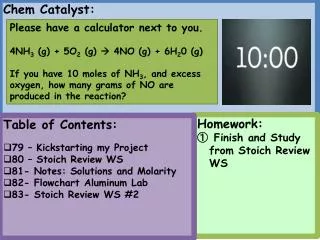
Today’s Agenda: • Do Now • Notes- Writing half reactions • HW: Practice Worksheet
Half Reactions: • Show only oxidation or reduction • Include electrons gained or lost to balance charges • Losing electrons (oxidation): electrons appear as a product • Gaining electrons (reduction): electrons appear as a reactant
Half Reactions: • Add electrons to the following half reactions. Al Al3+ S2- S Cl Cl- Sr Sr2+
Voltaic cell • Consists of 2 half-reactions • Cathode: where reduction occurs (RED CAT) • Anode: where oxidation occurs • Electrons flow from anode to cathode (electric current)
Cu/Zn Redox Reaction • Occurs spontaneously • E- flow from Zn to Cu • This flow of electrons creates an electrical current • http://www.youtube.com/watch?v=V7fS1QofMqI
Voltaic Cell Summary: • Electrons are given off when zinc is oxidized (anode) • E- pass through wire to copper • Copper is reduced (cathode) • Ions move across salt bridge http://www.youtube.com/watch?v=nNG5PMlHSoA&feature=related