Catalyst
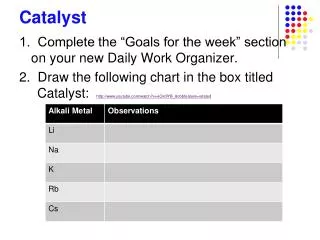
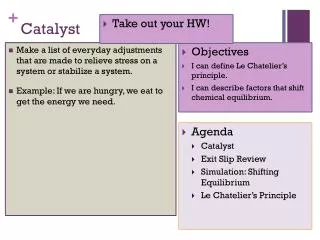
Catalyst . 1. Complete the “Goals for the week” section on your new Daily Work Organizer. 2. Draw the following chart in the box titled Catalyst: http://www.youtube.com/watch?v=eCk0lYB_8c0&feature=related. Objectives. I can define ionization energy, atomic radius, and electronegativity .

Catalyst
E N D
Presentation Transcript
Catalyst 1. Complete the “Goals for the week” section on your new Daily Work Organizer. 2. Draw the following chart in the box titled Catalyst: http://www.youtube.com/watch?v=eCk0lYB_8c0&feature=related
Objectives • I can define ionization energy, atomic radius, and electronegativity. • I can identify and explain trends in ionization energy, atomic radius, and electronegativity.
Agenda • Catalyst • Periodic Trends Lab • Periodic Trends Post-Lab Presentations • Exit Slip: Completed Post-Lab
Periodic Table Trends A trend is a predictable change in a certain direction. Understanding periodic trends allows us to make predictions about the chemical behavior of the elements.
Periodic Trends… • We already know: • Atomic number increases as we move down a group and also increases as we move across a period. • Trend from metals to semimetals to nonmetals as we move across a period from left to right. • We just learned: Reactivity of alkali metals increases as we move down the group. • Today’s investigation is about the trends regarding: • Ionization energy • Atomic Radius • Electronegativity
Pre-Lab Notes: • What is Ionization Energy? • It is the energy required to take electrons away from an atom. • What is Atomic Radius? • It is the size of the atom. • What is Electronegativity? • It is how much an atom pulls electrons that it is sharing in a chemical bond towards its nucleus.
Investigation Procedure • Safety Precaution: Do NOT touch the metal tip of the hot glue gun or the hot glue at any time. There is a very limited supply of glue, scissors, and straws so please use these materials carefully.
Materials • Straws • Scissors • Periodic table • Hot glue gun • Periodic trend data chart • Ruler
Procedure Overview • Follow directions on your trend data to construct your straw model. (25 min) • Determine the direction of your trend and add this information to your model. (3 min) • Clean up work space. (3 min) • Complete the Post-Lab questions. (20 min) • Be ready to present your trend to the class. (25 min)
Attraction and Repulsion of Electrical Charges + - - - + + Particles with unlike charges attract one another. Particles with like charges repel one another.
Ionization energy Ionization energy decreases Ionization energy increases
Ionization Energy Trend:Why? • Decreases as you move down a periodic table group because • the valence electrons are farther from the nucleus • and there are more electrons in the inner energy levels shielding the attractive charge of the nucleus. • Increases as you move across a period because • The number of protons increases but the valence electrons are in the same energy level. This creates more nuclear force holding the electrons in.
Ionization energy: The amount of energy needed to remove an electron from an atom or ion. Atoms that want to gain electrons have high ionization energies. Atoms that want to lose electrons have low ionization energies. Noble gases have the highest ionization energy.
Atomic radius increases Atomic radius: the size of an atom Atomic radius decreases
Atomic Radius Trend: Why? • Atomic radius increases as we move down a group because • Valence electrons are in a higher energy level. • Atomic radius decreases as we move across a period from left to right because • One more proton and one more electron are added as we move from left to right but the electrons stay in the same energy level. The added protons pull harder on the electrons, decreasing the size of the atom.
Electronegativity Electronegativity decreases Electronegativity increases
Electronegativity Trend: Why? • Electronegativity decreases as we move down a group because • Valence electrons are in a higher energy level and farther from the positively charged protons. • Electronegativity increases as we move across a period from left to right because • One more proton and one more electron are added as we move from left to right but the electrons stay in the same energy level. The added protons pull harder on the electrons, increasing electronegativity.
5) Electronegativity: the tendency of an atom in a molecule to attract shared electrons to itself. Elements that want to gain electrons have highelectronegativies. Elements that want to lose electrons have lowelectronegativies. exception: noble gases have no electronegativity