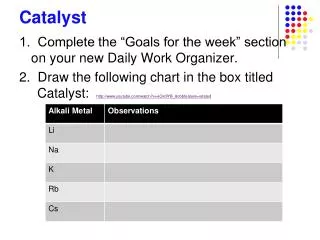
Catalyst
Catalyst. Calculators today! 1. What would be the new volume if the pressure on 8 L of gas is increased from 10 atm to 12 atm ? 2. You have 0.2 L of neon at a temperature of 289 K. If you the temperature was initially 350 K, what was the initial volume of the gas?.


Catalyst
E N D
Presentation Transcript
Catalyst Calculators today! 1. What would be the new volume if the pressure on 8 L of gas is increased from 10 atm to 12 atm? 2. You have 0.2 L of neon at a temperature of 289 K. If you the temperature was initially 350 K, what was the initial volume of the gas?
Gas Laws Practice • 1. What is Boyle’s Law? • 2. What is Charles’ Law? • 3. If I have 0.275 moles of gas at a temperature of 75 K and a pressure of 2.6 atmospheres, what is the volume of the gas?
Demo Day! • We will do a series of demonstrations and short labs to bring the gas laws to life! • After each one, you will explain what happened and WHY (using a gas law as proof)
Demonstrations • Egg in a Bottle • Can Crushing
Can you explain it? • A football inflated inside and then taken outdoors on a winter day shrinks slightly.
Can you explain it? • Hot air balloons increase slightly in volume as they rise higher in the air.
Can you explain it? • Car tires have a lower pressure in the morning than in the afternoon
Can you explain it? • A slightly underinflated rubber life raft left in bright sunlight swells up (Why shouldn't you overinflate your life raft when your ship goes down in tropical waters?)
Can you explain it? • Throwing a can of hair spray into a fire may cause it to explode.
Exit Slip 1. According to the ideal gas law, as the number of molecules goes up, the pressure will ___________. 2. According to Charles’ Law, as the temperature goes up, the volume will go ______________. 3. According to Boyle’s Law, as volume goes up, pressure will go ______________. 4. If I have an unknown quantity of gas at a pressure of 0.5 atm, a volume of 25 liters, and a temperature of 300 K, how many moles of gas do I have?