Chem Catalyst
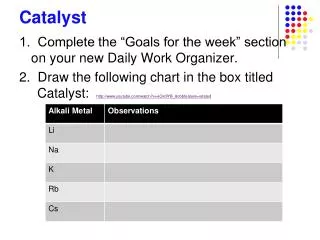
Chem Catalyst. Identify the type of reaction: Ag 2 S Ag + S 8 ZnS + AlP Zn 3 P 2 + Al 2 S 3 Cu + AgNO 3 Ag + CuNO 3 C 6 H 6 O 6 + O 2 H 2 O + CO 2 H 2 + O 2 H 2 O 2) Write a complete, balanced equation for the combustion of ethane (C 2 H 6 ). Today’s Agenda:. Catalyst

Chem Catalyst
E N D
Presentation Transcript
Chem Catalyst • Identify the type of reaction: Ag2S Ag + S8 ZnS + AlP Zn3P2 + Al2S3 Cu + AgNO3 Ag + CuNO3 C6H6O6 + O2 H2O + CO2 H2 + O2 H2O 2) Write a complete, balanced equation for the combustion of ethane (C2H6)
Today’s Agenda: • Catalyst • Recap: Identifying reactions • Quiz • Notes/Examples: Predicting products
Predicting Products: • Combination: • Form the ionic compound that results from the two elements • Ex: Write the equation for the reaction of solid Be and oxygen gas
Predicting Products: • Decomposition: • Separate the elements • Ex: Write the equation for the decomposition of HI
Predicting Products: • Single-Replacement: • A metal will replace a less active metal in the ionic compound • Ex: Write the equation for the reaction of solid iron and aqueous lead (II) nitrate
Predicting Products: • Double-Replacement: • Separate the ionic compounds and trade cations to write new ionic compounds • Ex: Write the equation for the reaction of NaOH and Fe(NO3)3