Microbial Control
Talaro Chapter 12. Microbial Control. Choosing a disinfectant or antiseptic…. often called “germicides”. -gaseous, liquid or solid state -effective concentration (versus toxicity) -broad spectrum? -low toxicity -penetration of surfaces of inanimate objects or tissues

Microbial Control
E N D
Presentation Transcript
Talaro Chapter 12 Microbial Control
Choosing a disinfectant or antiseptic…. often called “germicides” -gaseous, liquid or solid state -effective concentration (versus toxicity) -broad spectrum? -low toxicity -penetration of surfaces of inanimate objects or tissues -resistance to becoming inactivated by organic matter -noncorrosive or nonstaining properties -odor -affordability -availability
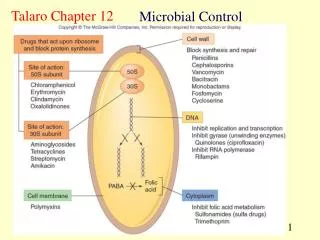
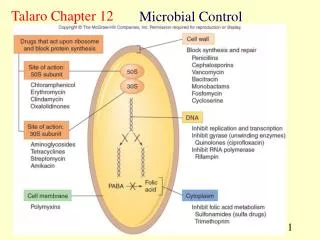
Chemical Decontamination Procedures 3 Levels • High - kill endospores… are sterilants – necessary for medical devices etc. e.g., catheters (some parts are not autoclavable) • Intermediate – Kills fungal spores but not endospores and generally kills most pathogens –used to disinfect items that touch mucus membranes but are not invasive • Low – Kills only vegetative cells of bacteria and fungi… probably kills most pathogens – for cleaning furniture, straps, electrodes… things that touch the skin surface
Chemical Groups of Germicides 1) Halogens (a group of non-metallic elements– fluorine, bromine, chlorine, and iodine Antimicrobial in the non-ionic state… Fluorine and bromine are dangerous so only Cl and I are used routinely Bacteriocidal, bacteriostatic, and even sporicidal with long contact time Cl and I are the active ingredients in over 1/3 of all antimicrobials marketed
Chemical Groups of Germicides (cont.) Cl: used for almost 200 years Cl2 gas, OCl- (hypochlorite) and NH2Cl (chloramine) – all react with water to form hypochlorous acid (HOCl) – reacts with certain amino acids and denatures proteins Disadvantages- relatively unstable… light, alkaline pH, and organic matter affect stability
Chemical Groups of Germicides (cont.) I: Penetrates cells of microorganisms well 2-3% in water or 70% alcohol used a topical antiseptic for surgery 5-10% for Iodophores- I complexed to a polymer (e.g., polyvinylalcohol)… solves several problems… -allows for slow release of I -increases penetration -less staining and irritating e.g., Betadine
Chemical Groups of Germicides (cont.) 2) Phenol derivatives (carbolic acid) – broad spectrum -derivatives referred to as phenolics - toxic to host cells – Lister (antiseptic surgery) – so not used much as antiseptics – disrupts cell walls, membranes and denatures enzymes - all antimicrobials compared to phenol still (the phenol coefficient) -phenol used rarely but low percentages in soap solutions are commonly used (e.g., 1-3%, Lysol) - hexachlorophene - triclosan (Safeguard soap)
Chemical Groups of Germicides (cont.) 3) Chlorhexidine – organic molecule with two phenol rings and Cl - targets cell membranes and dentures enzymes - bacteriocidal for Gram- and Gram+ organisms but is not sporicidal - low toxicity – used as obstetric antiseptic neonatal wash, wound antispetic, mucus membrane irrigant, preservative in eye solutions, and handscrubbing for doctors, preparing skin for surgical procedures
Chemical Groups of Germicides (cont.) 4) Alcohols - -OH functional groups - ethyl and isopropyl are the only suitable candidates • Concentrations of >50% dissolve lipids • Dentures proteins as well in cytoplasm • Optimal concentration is 70% (water is required for proteins to coagulate) • Removes oils on skin in which bacteria may be embedded • Disadvantages – evaporates quickly, some organisms can use it, some toxicity with isopropyl
Chemical Groups of Germicides (cont.) 5) H2O2 and related compounds - bacteriocidal (broad spectrum) and even sporicidal at high concentrations - action is through the hydroxyl free radical (OH-) - 3% H2O2 (aqueous) is routinely used for skin and wound cleansing and mouthwashing - can be a sterilant at 35%... Can get into parts of medical devices without corrosion - can be vaporized - Ozone (O3) can also be used as a disinfectant and works similarly to preoxide
Chemical Groups of Germicides (cont.) 6) Detergents and soaps Detergents – have surface action (called surfactants) – are polar, charged molecules - most soaps fit here - cationic compounds work better than anionic ones… all have a long uncharged hydrocarbon chain (allows detergent to disrupt the cell membrane) - best example – quaternary ammonium compounds (quats) - benzalkonium chloride, cetylpyridinium chloride are commonly used - mixed with cleaning agents, used to disinfect floors, furniture, equipment surfaces… not good enough for medical devices (level of disinfection is low) - high concentrations – effective against some Gram+, viruses, fungi, and algae Low concentrations, mainly bacteriostatic
Chemical Groups of Germicides (cont.) Soaps: made up of fatty acids of oils with sodium or potassium (are salts of FA) - Only weakly microbicidal and in fact, many bacteria including Pseudomonas aeruginosa can live and grow in soap dishes - Removes oil on skin however and can be very effective at removing bacteria on skin if scrubbing accompanies the washing - Thus many soaps add some other compound like I or chlorhexidine
Folk Medicine (pre 1890’s) Plant products Opium/morphine/heroin Quinine Caffeine Cocaine Salicin Digitoxin Diptheria antitoxin (1891) Salvarsan for syphillus (1911) Arsenic compound Paul Ehrlich “magic bullets” Penicillium inhibited streptocci on petri plate (1928) Alexander Flemming Protonsil Red (1935) Streptococci Gerhard Domagk Sulphanomide Penicillin (1941) Florey & Chain Streptomycin (1944) Effective against tuberculosis Selman Waksman Streptococcus griseus History of Chemotherapy
Chemical Groups of Germicides (cont.) 7) Heavy Metals – Hg, Ag, Au, Cu, Zn, As have all been used at one time or another for microbial control - Most too toxic to host!!! And can be absorbed through skin so even tough to use as disinfectants…may cause allergic reactions - broad spectrum as they bind and inactivate proteins - Mercurochrome - AgNO3 Solutions and newborns – are still some Ag-containing ointments particularly for burns Some other disadvantages – Microbes can develop resistance to metals (genes for this often times on the same plasmids as those that have R factors!) - Large amounts of organic material (host wastes etc.) neutarlize action - Age generally weak as to their antiseptic qualities
Chemical Groups of Germicides (cont.) 8) The aldehydes – formaldehyde, and glutaraldehyde (have –CHO functional group) - Intermediate to high level disinfectants - “fixes” proteins- preserves tissues for many years… - Very toxic - Glut can be even used as a sterilant… killing spores in about 3 hours - “chemiclave” - vaporized glut - Formaldehyde gas – formalin (37% aqueous solution) – acts more slowly than glut - Formalin is a carcinogen
Chemical Groups of Germicides (cont.) 9) Gaseous sterilants and disinfectants • Ethylene oxide- • Propylene oxide – • Chlorine dioxide -
Chemical Groups of Germicides (cont.) 10) Dyes – even bacterial stains - Crystal violet -Malachite Green 11) Acids and Bases – - Acetic acid – 12) Salt (NaCl) -
Remember: • Antiseptic and Disinfecting • Activity is: • Concentration (of compound) • Time • Organism • Dependent!!!
Microorganisms & Antimicrobial Drugs • Antibiotics are common metabolic products of aerobic bacteria & fungi • Streptomyces (an Actinomycete)& Bacillus • Penicillium & Cephalosporium (fungi) • Inhibit other microbes in the same habitat, antibiotic producers have less competition for nutrients & space
Mechanism of Antimicrobial Action • Antimicrobial drugs may simply inhibit bacterial growth • BACTERIOSTATIC • Drugs may also actively kill bacteria - BACTERIOCIDAL • All exert action by inhibiting particular aspect of cellular • physiology • Five primary aspects of cellular physiology are targets • Cell Wall Synthesis • Protein Synthesis • DNA/RNA Synthesis • Maintenance of Plasma Membrane • Synthesis of Essential Metabolites
Selective Toxicity Essential Metabolite
Inhibition of Cell Wall Synthesis Peptidoglycan b-lactam Weak points in peptidoglycan Bulging surface of cocci
Page 352 b-lactams BSCI 424 PATHOGENIC MICROBIOLOGY U of Maryland
N-acetylmuramic acid (NAM) Peptidoglycan • Macromolecule composed of a repeating framework of long chains cross-linked by short peptide fragments • Unique to Bacteria • Composed of 2 sugars: NAG & NAM • Sugars alternate in the backbone • Rows linked by polypeptides • Provides strong, flexible support to keep bacteria from bursting or collapsing because of changes in osmotic pressure N-acetylglucosamine (NAG)
Intact peptidoglycan b-lactam antibiotics block peptidases that link the cross bridges between NAMs NAM & NAG glycans cross linked by peptide bridges Penicillins Cephalosporins Relatively safe since mammals do not have peptidoglycan
Broad spectrum penicillins and cephalosporins can cross the cell walls of Gram negative bacteria. Carbenicillin & ceftriaxone Some penicillins are less effective against Gram negative bacteria. Some cannot penetrate the outer membrane well.
Penicillins • All consist of 3 parts • Thiazolidine ring • b-lactam ring • Variable side chain dictates microbial activity • R
Cephalosporins • Account for majority of all antibiotics administered • Cephalosporium acremonium • b-lactam • Relatively broad-spectrum • Resistant to most penicillinases & cause fewer allergic reactions • Some are given orally, many must be administered parenterally • Other than via the digestive tract • Intravenous or intramuscular injection
Vancomycin • Streptomyces orientalis • Disrupt alanine-alanine bridges that link NAM in most Gram + bacteria • Bacteria lacking alanine-alanine bridges are resistant publications.nigms.nih.gov/.../chapter1.html
Bacitracin • Peptide antibiotic • In the product Neosporin • Bacillus subtilis • Blocks movement of NAG and NAM from the cytoplasm • Effective against Gram + • Topical antibiotic preparations • Bacitracin has a high toxicity which precludes its systemic use
b-lactams, vancomycin and bacitracin inhibit bacteria from building peptidoglycan • Have no effect on already existing peptidoglycan • Thus really most effective against actively growing cells • Dormant cells or endospores are not susceptible
Inhibitors of Specialized Cell Wall Synthesis • Unusual cell walls - Mycobacterium tuberculosis • Isoniazid inhibits synthesis of mycolic acids… acid-fast stain… • Integral part of waxy cell wall • Essential for cell wall assembly • Ethambutol inhibits incorporation of mycolic acid into waxy layer • Both extremely effective against Mycobacterium • Mycobacteria have generation times of 12 – 24 hours (slow growers) • Drugs administered over many months
Outer lipids Mycolic acid Lipoarabinomannan Cell wall skeleton Arabinogalactan Peptidoglycan Plasma membrane Mycolic acids make up much of the cell wall of Mycobacterium es.wikipedia.org/wiki/Imagen:Mycobacterial_cell_wall_diagram.png
Drugs that Block Protein Synthesis • Ribosomes of eukaryotes differ in size and structure from prokaryotes - 80s versus 70s – don’t forget subunit differences… • Antimicrobials selectively target bacterial translation • Some of these drugs damage ribosomes in the eukaryotic mitochondria or chloroplast • Contain a prokaryotic type of ribosome
Most of These Effect Some Part of Translation • Aminoglycosides • Insert on sites on the 30S subunit and interfere with the reading of the codons on the mRNA • Tetracyclines • Block attachment of tRNA on the A acceptor site and stop further synthesis • Chloramphenicol • Attach to the 50S subunit • Prevents formation of peptide bonds • Macrolides & Lincosamides • Attach to the 50S subunit • Inhibit translocation • Movement of ribosome from one codon to another • Oxazolidinones • Inhibits initiation complex formation • Binds to the 50S subunit and prevents the 30S complex from forming the 70S complex
Antibiotics That Interfere With Protein Synthesis Aminoglycosides – Gram- aerobic bacteria -Pseudomonas, Enterobacter, even Mycobacterium… - not well absorbed through gut so usually administered intravenously and intramuscularly Examples: Streptomycin, Neomycin, Gentamycin, Kanamycin
Antibiotics That Interfere With Protein Synthesis Tetracyclines: - Broad spectrum - First one was chlortetracycline (aureomycin) by Streptomyces aureofaciens… then Oxytetracycline by S. nimosus - Naturally-occurring versus synthetic • Generally low toxicity… but Ca2+ problem… absorbed well though gut • In 2005 tigecycline was developed… a new class of antibiotics called the glycylcyclines… first new tetracycline antibiotic in over 20 years… new ones are in clinical trials now • Examples: Synthetic- Doxycycline, Minocycline
Antibiotics That Interfere With Protein Synthesis Macrolides - broad spectrum • Used for upper resp. and soft tissue infections • Used often as substitute for penicillin • Toxicity low- absorbed well through gut • Examples- erythromycin, azithromycin (Zithromax or Z-Pak)
Antibiotics That Interfere With Protein Synthesis Lincosamides – action like macrolides • Lincomycin from S. lincolnensis • Used to treat Staph and Strept infections and also good against some anaerobes, and protozoans such as Plasmodium (malaria) • Other examples- Clindamycin (basically replaced Lincomycin) • Clindamycin usually give intravenously
Antibiotics That Interfere With Protein Synthesis Oxazolidinones – Good against Gram+’s - These were developed in the 90’s and used against MRSA when Vancomycin failed… - First one developed was Linezolid (Zyvox)… - Excellent oral bioavailability… and can be injected as well - Other examples: AZD2563 (AstraZeneca)…shows great promise….
Drugs that Disrupt Cell Membrane Function • A cell with a damaged membrane dies from disruption in metabolism or lysis • These drugs have specificity for a particular microbial group • Based on differences in types of lipids in their cell membranes. • Polymyxins • Interact with phospholipids and cause leakage • Originally the only class of antibiotic for Gram - bacteria • Not effective against Gram + bacteria • Topical use • Toxic to kidney cells
Pyrazinamide • May inhibit fatty acid synthesis • Transport across the cytoplasmic membrane is disrupted • Uniquely effective against Mycobacterium • Mycobacterium accumulates this drug
Inhibition of an Essential Metabolite • Sulfonamides and trimethoprim interfere with folate metabolism • Block enzymes required for tetrahydrofolate synthesis needed for DNA & RNA synthesis • Competitive inhibition by sulfonamides • Structural analog • Drug competes with normal substrate for enzyme’s active site • para-aminobenzoic acid (PABA) • Trimethoprim inhibits dihydrofolate reductase • Next step in the tetrahydrofolate acid biosynthetic pathway • Synergistic effect • Both drugs administered together • An additive effect, achieved by multiple drugs working together, requiring a lower dose of each