Understanding Ternary Acids: Composition, Naming, and Examples
Ternary acids, also known as oxyacids, are composed of three elements, typically including hydrogen as H⁺ and a non-metal with oxygen. Recognizable by the formula H.Nonmetal.O, common examples are nitric acid (HNO3), phosphoric acid (H3PO4), and sulfuric acid (H2SO4). These acids usually end with the suffix "-ic" and do not use the prefix "hydro" as they are not binary. Understanding their structures and names, based on group models from the periodic table, helps in mastering chemical nomenclature.

Understanding Ternary Acids: Composition, Naming, and Examples
E N D
Presentation Transcript
Nomenclature Tertiary (or Ternary) Acids
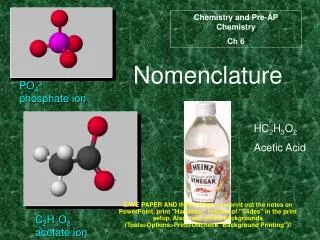
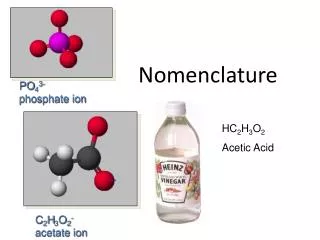
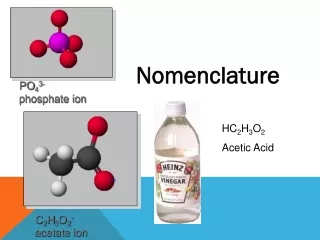
Ternary Acids • Ternary = 3 elements in compound • Acid = Hydrogen usually present as H1+ ion • Also called “Oxyacid” because oxygen also present • Recognize formula as H Nonmetal O • Examples: HNO3 H3PO4
6 Model Ternary Acids • Memorize these 6 example formulas and names on periodic table to represent groups 13-17 13 14 15 16 17 H3 B O3 H2 C O3 H N O3 Boric Carbonic Nitric acid acid acid H3 P O4 H2 S O4 H Cl O3 Phosphoric Sulfuric Chloric acid acidacid
Naming Ternary Acids • Notice all these example acids ended with suffix –ic • There is noprefix “hydro” because not binary acid • Notice the exact number of oxygens in each model for each group • Group 15 follows Phosphoric acid model
Your Turn to Try • Write the name and formula for each element based on the group model • Ge • Br • Se • As
System of Varying Oxygen • Study the pattern of how prefix and/or suffix change as the number of oxygens in the formula change H2CO4 Percarbonic acid Added extra oxygen H2CO3 Carbonic acid Model formula 3 oxygens H2CO2 Carbonous acid Removed 1 oxygen H2CO Hypocarbonous acid Removed 2 oxygens
Your Turn to Complete Chart • Write all possible formulas and names for the elements N and S
Your Turn to Complete Chart • Write all possible formulas and names for the elements Si and Te