The Evolution of the Periodic Table: Mendeleev to Modern Science
In the early 1860s, scientists recognized over 60 elements but lacked organization. Dmitri Mendeleev made history in 1869 by categorizing these elements based on properties like density and melting point, arranging them by increasing atomic mass. His groundbreaking periodic table revealed a repeating pattern of elemental properties, allowing for predictions of undiscovered elements. Following Mendeleev, Henry Moseley redefined the table by atomic number, emphasizing that properties change periodically with atomic numbers. This system categorizes elements into metals, nonmetals, and metalloids, establishing the foundation for understanding chemical behavior.

The Evolution of the Periodic Table: Mendeleev to Modern Science
E N D
Presentation Transcript
Early 1860’s • Scientists knew some of the properties of more than 60 elements that existed. • BUT…..they were not in any order.
Demitri Mendeleev 1869 • Mendeleev was the first scientist to write the names and properties of each element on the an index card. • He used properties such as; density, appearance, and melting point. • He then arranged the elements in order of increasing atomic mass. • Mendeleev discovered a repeating pattern that took place among the elements going from left to right across and up and down.
Demitri Mendeleev 1869 • Periodic – means happening at regular intervals, “repeating”. • Example – the days of the week. • He left spaces or question marks for unknown or undiscovered elements. • Mendeleev’s arrangement of elements became known as the periodic table of elements.
Henry Moseley 1914 • Moseley – a British scientist, determined the atomic number (# of protons) in an atom. • Periodic Table is also arranged by increasing atomic number or by increasing number of protons. • Periodic Law – the repeatingchemical and physical properties of elements change periodically with the element’s atomic numbers.
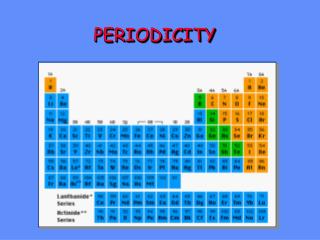
Periodic Table • A periodic table is an arrangement of elements in columns, based on a set of properties that repeat from row to row. (page 338-339)
Periods are the rows in the periodic table (horizontal). • Elements change from metals to nonmetals as you move from left to right across the period. • Groups/Families are the columns on the periodic table. (vertical) • Properties of elements repeat in a predictable way. (Periodic Law) Group -- Period ------------
Classes of elements • Elements are classified as metals, metalloids, and nonmetals based on their properties. • Metals – Start at the far left hand side and stop at the zigzag line. • - Most elements are metals. • - Most are solids at room temperature. • - The exception: Mercury (Hg) • - Metals tend to be shiny, luster. • - Malleable – hammered into thin sheets. • - Good conductors of electricity/heat. • - Ductile – made into thin wires.
Classes of elements • Nonmetals – Start at the far right hand side and stop at the zigzag line. • - More than half are gases at room temperature. • - Nonmetals tend to be dull, lack luster. • - Brittle –will shatter if struck with a hard object. • - Poor conductors of electricity/heat - insulator • - Non-ductile – cannot be drawn into wires.
Classes of elements • Metalloids – elements that border the zigzag line. • - Tend to have properties that are in between metals and nonmetals. • - Called semiconductors Tellurium is shiny, but it is brittle and can easily be smashed into a powder. Boron is very brittle. At high temperatures it is a good conductor of electricity/heat.
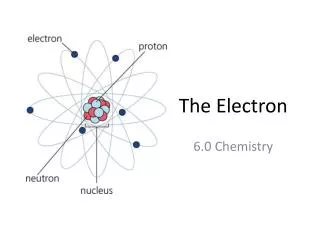
Valence Electrons • A Valence Electron is an electron that is in the highest occupied energy level of an atom. • Elements in a group have similar properties because they have the same number of valence electrons.
Alkali Metals family • Group 1 • Mostreactive metals • Reactivity increases from the top to the bottom. • Francium is the most reactive metal! • So reactive many are kept under oil to prevent reacting with water or oxygen. • Found in nature only in a compound. • One Valence Electron • Give away one valence electron and form +1 ion in order to become stable.
Alkaline Earth Metals Family • Group 2 • 2 Valence Electrons • Give away 2 valence electrons and form +2 Ions in order to become stable. • Less reactive than group 1. • Magnesium used in photosynthesis. • Calcium used in teeth and bone.
Transition Metals • Groups 3 – 12 • 1 or 2 valence electrons • Less reactive than alkaline earth metals • Shiny, good conductors of heat and electricity. • Their properties vary widely. Iron is very reactive and can react with oxygen in the air to form rust. Many are solids at room temp, except Mercury. Many are silver in color except Gold. Titanium not very reactive.
Transition Metals Lanthanides and Actinides • Transition metals from periods 6 and 7. • Lanthanides are shiny reactive metals. • - Some of these elements are used to make steel. • Actinides are radioactive, or unstable. • - Elements listed after Plutonium, element 94 do not occur naturally in nature.
Boron Family • Group 13 • 3 Valence electrons • Give away 3 valence electrons and form +3 ions in order to become stable. • 1 metalloid (Boron) • Five metals • Aluminum is the most abundant metal in the Earth’s crust.
Carbon Family • Group 14 • 4 Valence Electrons • Can give away 4 or take 4 valence electrons and form +/- 4 Ions in order to become stable. • 1 Nonmetal • 2 Metalloids • 3 Metals • With the exception of water, most of the compounds in your body contain carbon.
Nitrogen Family • Group 15 • 5 Valence Electrons • Take 3 valence electrons and form -3 Ions in order to become stable. • 2 nonmetals • 2 metalloids • 2 Metals • Nitrogen and Phosphorus are used in fertilizers.
Oxygen Family • Group 16 • 6 Valence Electrons • Take 2 valence electrons and forms -2 Ions in order to become stable. • 3 nonmetals • 1 metalloid • 2 metal • Oxygen is the most abundant element in the Earth’s Crust.
Halogen Family • Group 17 • 7 Valence electrons • Take 1 valence electron and form -1 Ions in order to become stable. • Most reactive nonmetals • Reactivity increases from bottom to top. • Fluorine is the most reactive nonmetal.
Noble Gas Family • Group 18 • 8 Valence Electrons • Un-reactive (Do not form Ions) because they are stable. • The Noble Gases are odorless and colorless. • Used in light bulbs (Argon). • Make balloons float (Helium). • Used to make neon lights.